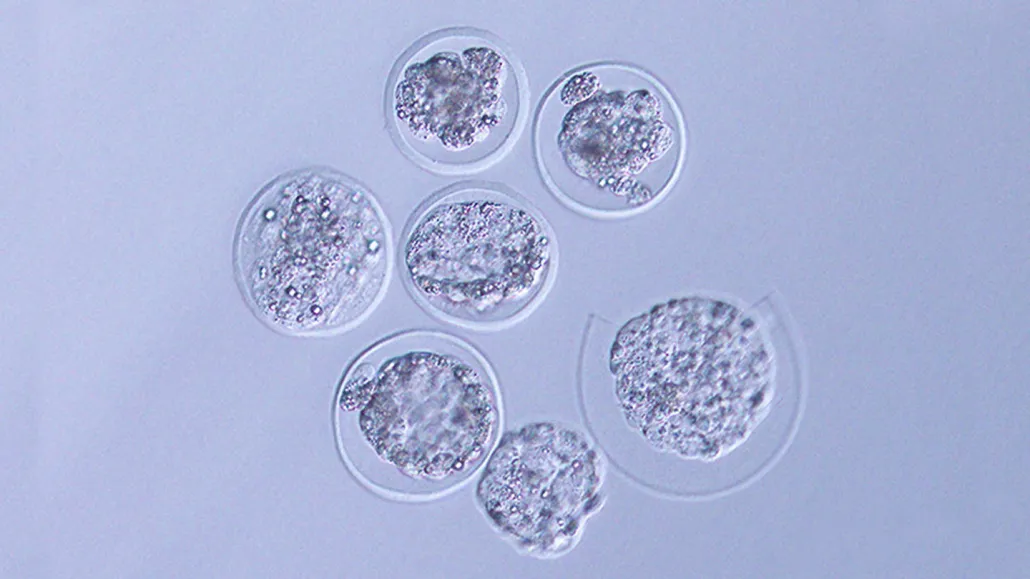
Two-cell mouse embryos cultured on the International Space Station and returned to Earth formed blastocysts (some shown).
S. Wakayama et al/iScience 2023
Mouse embryos can make it to an early stage of development in space.
In an experiment conducted in 2021, a few hundred frozen two-cell embryos from mice thawed and grew over four days on the International Space Station. Of the several dozen embryos that made it back to Earth, nearly a quarter formed healthy clusters of cells known as blastocysts.
The finding suggests that the radiation and weightlessness of space might not pose immediate obstacles to mammalian reproduction, researchers report October 27 in iScience.
The new study isolates only one part of the complicated process of reproduction and development. A blastocyst typically forms after fertilization and implants in the uterus before developing into the placenta and fetus.
But the result provides a starting point for biologists, says Christiane Hahn, a space biologist at the European Space Research and Technology Centre in Noordwijk, Netherlands, who wasn’t involved in the research. Mouse embryos are the first mammal embryos that researchers have grown in space, an important step in understanding how space affects human reproduction. Other experiments have involved animals such as salamanders, rice fish and quail.
Previous research suggested that the conditions in space are particularly harmful to the early stages of reproduction in mice. When in space, the animals have been too stressed to mate, and studies of the rodents’ eggs showed that they accumulated several mutations due to the heavy radiation. Freeze-dried mouse sperm, however, did remain viable after one six-year stint on the space station (SN: 6/11/21).
To overcome these challenges, biologist Teruhiko Wakayama and colleagues launched two-cell embryos to the ISS. A device specially made for the experiment held the fragile, developing cells. After the experiment ended, the cells were then sent back to Earth for analysis. Out of 360 samples, 72 survived the trip, and 17 of those developed into normal blastocysts. The undamaged cells successfully multiplied and took on new identities as either precursors of fetal tissue or the placenta.
Future alterations to the procedure could potentially increase the success rate, says Wakayama, of the University of Yamanashi in Kofu, Japan. The absence of perfectly sterile conditions in the space experiment probably exacerbated cell death, he says.
Blastocysts cannot survive for long outside of uteruses, so the experiment was designed to last only a few days. It’s unclear how the cells would fare as development unfolds. In future experiments, the team would like to implant any viable embryonic cells from space experiments in mice to find out.
The researchers are especially interested in further testing the impact of microgravity on the positioning of different cells in a blastocyst. The cells that give rise to the fetus usually all sink to the bottom of the cluster, encased by cells that will become placenta. Were the fetal cells to split into two spots instead, they’d create identical twin mice. Were they to split into more, the blastocysts would probably become unviable.
In the new study, three-quarters of the fetal precursors appeared to settle in the right location. But more research is needed to fully understand the effect of microgravity on the developing cells.






