The term “bar fight” does not actually appear in Saila Varis’ recent paper in the journal Trees or in her Ph.D. dissertation on the Scots pine. But she’s a good sport about discussing whether her research suggests that tree pollen grains have their own versions of nose-punching brawls over female favor.
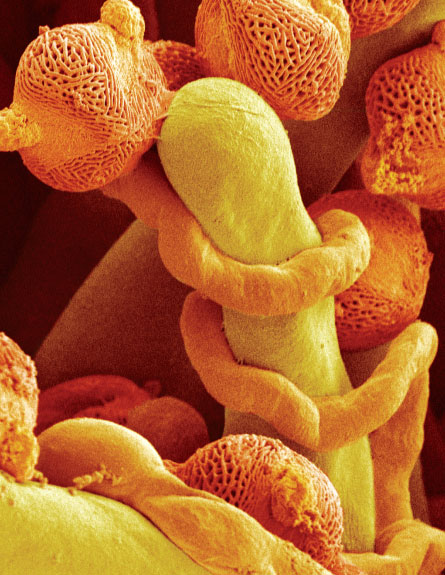

After all, pollen grains from genetically different trees of the same species appear to be able to sabotage each other’s race to a mate, says Varis, of the Finnish Forest Research Institute near Helsinki. Though it is not exactly like a bar fight, she says, there are hints of male-versus-male competition.
Plant pollen may be basically microscopic dust, but as far as evolutionary biology goes it can be as male as any swaggering pool-hall hound with smooth moves and high hopes for the night. Pollen grains competing for access to the alluring green nubbins of female tissue in a pine tree add to growing evidence that a quirky evolutionary force known from animals, called sexual selection, may also show up in plants.
This evolutionary force is not the familiar survival of the fittest, but rather a sort of survival of the sexiest. For no matter how vulnerable a feather display or antler rack may make its bearer, the features can count as an evolutionary advantage if they boost the organism’s number of offspring. Biologists have plenty of examples of such sexy evolution among animals. Finding examples among plants has been a trickier business, though.
What’s a loud, visible chair-smashing fight between animals could in plants be a silent molecular conflict of pollen grains, a seepage of compounds or the activation of genes. Scientists are now developing ways to scrutinize these hidden interactions to look at basic questions of plant sexual selection. Evolutionary biologists want to know whether individual plants can exercise some choice among possible mates, and whether those mates vie among themselves. Though the means, the scale and the potential for lethal spurs or extreme lipstick are quite different among plants, the pressures driving flirting and fighting may be the same.
Beastly start
Even though “survival of the fittest” has become the bumper sticker explanation for how natural selection shapes life, the phrase omits a crucial point: An aardvark or a zebra could grow so robust, so attuned to its local environmental challenges, that it might in theory live forever — yet it could still end up as an evolutionary dud. An organism not only has to survive, but it also has to make babies. Otherwise it’s as much of a dead end as a weakling culled from the gene pool at birth.
Darwin acknowledged that survival was not the whole story. In the same chapter of The Origin of Species that describes natural selection, he introduced sexual selection, which he describes as depending on “a struggle between the individuals of one sex, generally the males, for the possession of the other sex.” He observed that reproductive success doesn’t depend just on robust vigor; rooster spurs and stag antlers trounce rivals, and extravagant feathers dazzle.
As the idea of sexual selection developed, theorists predicted that these special devices for winning a mate could drain resources that would otherwise go to the business of survival.
Darwin’s ideas on sexual selection could make sense of why the males and females of a species look different. Applying the principle to plants jolted some researchers because many plants grow as hermaphrodites, not as boys and girls with different features.
“There has of course been some reluctance to accept that sexual selection can operate in hermaphrodites, but I think the evidence is now clear that it can,” says plant biologist Spencer Barrett of the University of Toronto.
Theorists have gotten over the hurdle of seeking male or female individuals by considering male and female functions separately, even when they occur on the same plant. The male function produces little grains of pollen carrying the genetic material for fertilization. They travel, often hitchhiking on animals or wafting in wind, to a receptive surface on a female organ, called a pistil. Once there, the pollen grains can fertilize the ovules, or plant eggs, tucked inside.
For a plant-friendly definition then, in a 2011 review in Current Biology, John Pannell of the University of Lausanne in Switzerland and Jamie Moore of the University of Oxford in England de-emphasize separate males and females and describe sexual selection in terms of “a process that acts to increase mating success.”
To clarify, this research field is not just the familiar and huge endeavor to understand how flowers lure, manipulate and even betray the animals carrying pollen from he parts to she parts. Tactics of pollination have a long and distinguished tradition of study dating back to the Big D himself. Despite these glorious shenanigans of blooms and birds and bees, plant sex is really plant-to-plant.
And plants do appear to have some say in what other plants they knock boots with, potentially boosting their mating success. When birds and bees brush pollen onto the receptive surface of female flower parts, scientists have found, the seeds that get sired do not always directly reflect what the pollen express dropped off. This “nonrandomness” of mating suggests some form of influence is at play.
In greenhouse experiments with a California wildflower called Chinese houses (Collinsia heterophylla), evolutionary plant biologist Åsa Lankinen of the Swedish University of Agricultural Sciences at Alnarp has found that the first pollen to arrive on the female part of the flower often has an advantage in siring seeds. Yet there appear to be exceptions. When pollen from four particular donors was applied at different times, the final fatherhood turned out unexpectedly, Lankinen and Josefin Madjidian of Lund University reported in the July American Journal of Botany. Pollen grains that researchers delivered a day later than other grains still ended up siring up to 47 percent of a particular plant’s seeds.
How this works remains a puzzle. Perhaps the female part of the plant is somehow choosing pollen the way a peahen chooses among peacocks. Or certain pollen may compete more successfully to sire seeds. Pegging just one of these possibilities hasn’t been easy.
Choices, choices
Only a tiny (though colorful) minority of botanists use the word “brain” for a plant part. So in considering how sexual selection might work out in plants, “I think it was hard for people to see how a plant was able to choose,” Lankinen says. She, like many other biologists, instead starts with the basic attitude that choice doesn’t have to be conscious; it’s not clear whether even Homo sapiens truly thinks when undressing.
In many species, a female organ can reject pollen from other blooms on the same plant. Among those species are the Flanders poppy, flowering tobacco and field mustard, as well as the Oxford ragwort analyzed by Alexandra Allen of the University of Bristol in England and her colleagues in the September 2011 Annals of Botany. The female reproductive tissues in a bloom can recognize genetic factors signaling that a pollen grain is too closely related. And depending on the species, those factors may block or reduce self-fertilization.
Rejecting pollen isn’t the only way a plant could wield its influence, though. Female parts may be able to affect mate number and diversity too. Typically the female part of a flower does not need just one pollen grain, but a dusting of grains to sire the multiple seeds in her seedpod. That pollen may originate from one plant or many. In the sometimes alien green zone of plant sex, mating is not about finding Mr. Right Pollen Grain, but Mr. and Mr. and Mr. Right.
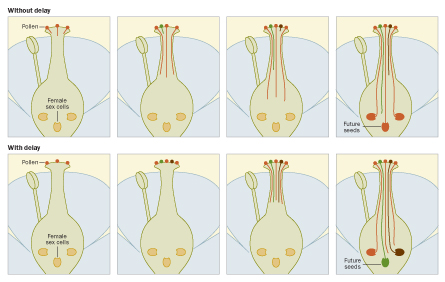
In the flowers that Lankinen studies, Chinese houses, the female pistil takes the usual form of a receptive surface, the stigma, atop a stalk that leads down to carefully wrapped and protected ovules at the pistil base. Pollen lands on the stigma and then sprouts tubes that grow downward through the plant tissue to reach the ovules. When a pollen tube arrives at an ovule, fertilization occurs.
But Chinese houses delay the start of the race, a timing twist proposed as a form of female choice. Even though the flower sits open and, it would appear, ready to go, pollen that lands on the stigma for the first day or so is just stuck sitting there. The female structure keeps elongating and bending until at last, the pollen grains are off. By instituting the delay, the stigma collects a variety of pollen, increasing the diversity rushing toward the ovules. In plant studies, Lankinen has found, a batch of pollen from multiple sires starting pollen tubes all at once led to more seeds from more sires than did comparable pollen delivered over two days.
The finding joins other examples that suggest that female plant parts may manipulate the fertilization process. In a classic study of wildflowers called anemones, the stigma’s surface begins turning receptive at the outer edges and matures in an inward wave. The final patch to allow pollen to start tubing toward the prize is the center, where pollen has the shortest distance down to the ovules. The pattern evens the odds for pollen that landed near the outer edge.
Once pollen tubes have reached and fertilized the ovules, some fertilized eggs wither instead of growing into seeds. Botanists have proposed that flowers may be selectively aborting certain embryos — possibly because they come from a loser sire.
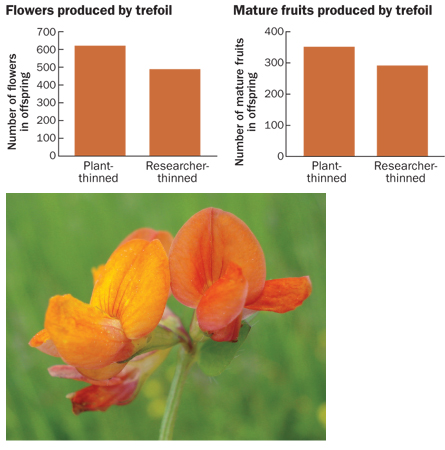
Barrett cites two classic studies he finds convincing: Small legumes called bird’s-foot trefoil (Lotus corniculatus) end up producing fewer seeds than the number of ovules fertilized. In an effort to explore whether such abortions happen randomly or might be a mechanism for weeding out unpromising offspring, Andrew G. Stephenson of Penn State University and his colleagues removed a random selection of flowers containing fertilized ovules so the ovules couldn’t develop. The fruits that formed on the plants allowed to abort their own way held more seeds with higher quality than did the fruits on plants that the researchers plucked. And with yellow-flowered desert perennials in the borage family (Cryptantha flava), natural processes of seed elimination produced better seeds than random laboratory processes did, Brenda Casper of the University of Pennsylvania has found.
Such research findings offer intriguing hints that choice may be involved. But since this is subtle, complex and mostly invisible biology, there’s a lot of cautioning and “we need more research” in presenting the progress so far.
That bar fight
If one plant sex can do some choosing, members of the other sex, whatever that means in a plant, may indeed compete or outright fight among themselves.
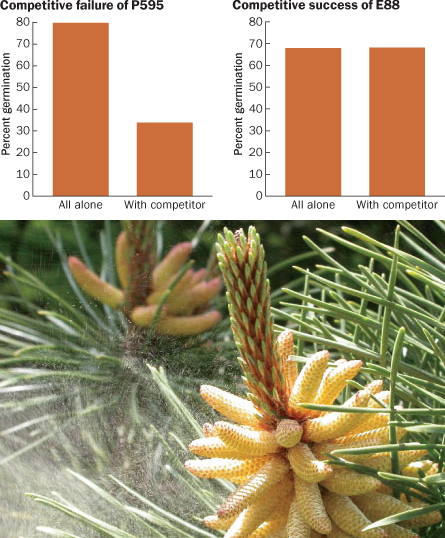
One of the most recent tests of the idea, with Varis’ Scots pines, pitted clones of trees from southern Finland against clones from the northern region. (This contest was part of a long-term project on tree breeding and adaptation to climate change.) In the lab, Varis sowed pollen from one clone or from two in little growth cups furnished with identical nutrients that substituted for female tissue. Because no actual female tissue was around to influence growth, variation among males reflected just the interactions of the males.
In some combinations, pollen from different trees germinated much as it did when set out alone. But other combinations seemed to pit pollen grain against pollen grain. Most dramatically, a particular northern clone’s germination rate ended up dropping 46 percentage points when the clone was paired with a southerner than when it was left to germinate alone.
Pine pollen grains travel by air instead of by insect, and a lucky pollen bit lands in a droplet of liquid exuded by the female organ. Once in the droplets, pollen grains release proteins in the course of absorbing moisture and preparing to fertilize the ovules. Perhaps, Varis speculates, some of these compounds might start molecular fights. Even if a female just accepts whatever pollen succeeds in reaching the right places, males may influence their mating success by sabotaging each other.
Male flowers of broadleaf arrowhead, a North American aquatic plant, appear to compete by showing off instead of sabotaging. The insect-pollinated Sagittaria latifolia separates its sex organs in different flowers, although sometimes the same individual plant grows both male and female flowers. In a survey of all variations, individual male flowers grow bigger than female ones, Barrett and two Toronto colleagues reported in the September Annals of Botany. Researchers looking at all the male flowers on a particular plant, single-sex as well as hermaphrodites, found that the male displays lasted longer than female displays, with new flowers opening as old ones finished blooming.
These sex differences in blooms fit the pattern of males competing with each other via greater advertising budgets to lure insects to spread pollen. The two sexes differ in form, like the stags and does that starred in Darwin’s original explanation of sexual selection.
Plants might even have their own versions of peacock tails. Demonstrating that an evolutionary force has driven one sex to such an impractical mating extravagance (iridescent feathers might attract predators, after all) isn’t easy, but a species in a plant genus that Pannell’s lab works on is under scrutiny as a candidate. A fire-adapted shrub from southern Africa has individuals with only male parts and others with only female ones.
When Leucadendron xanthoconus blooms, leaves at the tip of the branch near the actual flowers turn yellow as part of the show. The flowers and leaf displays of purely male plants can become so flamboyant that they shade parts of the regular green working leaves. (When flowering season ends, the shrub goes green again, much like a bird shedding its breeding plumage until the next season.) These male floral displays grow larger than females’ as if males are competing to spread more, more, more of their pollen.
The strongest suggestion that the shrubs may show signs of the kind of sexual selection seen in animals, though, comes from evidence that the competitive displays actually shorten male life expectancy, exacting a survival cost. Routine menaces such as fungus attacks kill males more readily than females, as if cutting back on regular green leaves in favor of flashier ones weakened the plants.
Pannell and his colleagues are still studying the female side of Leucadendron to try to figure out what evolutionary pressures may work on the female form. Some species keep their seeds snug inside cones on the mother plant for years if necessary until a fire cues the release, and this maternal care correlates with bigger differences between the sexes in Leucadendron species, Pannell’s team reported last year in the Journal of Ecology.
If the sexual landscape of plants turns out to show abundant signs of fights and flirting, the tensions could easily play out in another phenomenon familiar from animals: battles of the sexes. Researchers are starting to look at conflicts for resources between male and female functions in the same plant. After the pollen bash in the bar, there could be some slaps on the face and door slamming in the parking lot.







