Using a made-from-scratch genome, scientists have breathed a new kind of life into a bacterium. The feat, published May 21 in Science, holds great promise for creating designer organisms that might do things like produce vaccines, synthesize biofuels, purify water or eat spilled oil.
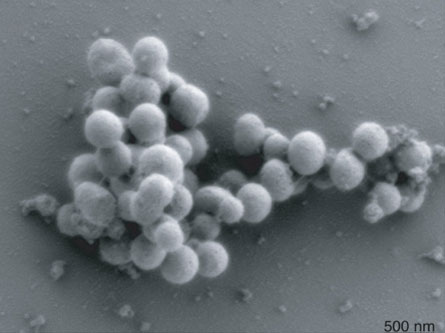
In the new study, researchers from the J. Craig Venter Institute carefully stitched together the entire genome of the bacteria Mycoplasma mycoides and put it into a different kind of bacteria, Mycoplasma capricolum. This unprecedented wholesale genome swap caused the M. capricolum cell to switch species. The newly converted cell was nearly identical to the natural M. mycoides.
“This was a proof of concept experiment showing that we could take the sequence out of a computer, build it and boot it up to make a synthetic cell,” says study leader Daniel Gibson of the Venter Institute’s campus in Rockville, Md.
This ability to transplant complete genomes from one species to another is “a marvelous piece of work,” says bioengineer James Collins, a Howard Hughes Medical Institute investigator at Boston University who was not involved in the study. “This represents an important advance for synthetic biology.”
Scientists at the Venter Institute already knew the DNA sequence of the M. mycoides genome. But they hadn’t been able to take the string of A’s, T’s, G’s and C’s stored in a computer and build the whole genetic instruction book in test tubes, put it in a cell and then show that it worked.
One of the major challenges in the new study was figuring out how to knit short pieces of DNA together in a particular order to create a large genome. Through earlier experiments, the team had found that proteins in yeast cells could quickly assemble large pieces of DNA. “We were amazed that yeast has this capacity, so we tried to push the limits,” Gibson says.
After going through three rounds of assembly in surrogate yeast cells with progressively bigger chunks of synthesized DNA, the researchers produced a record-setting genome that clocked in at 1,077,947 DNA letters. This synthetic genome was then introduced into M. capricolum cells, which began to forget their own characteristics and instead adopt the appearance and functions of the genome-donor species, M. mycoides.
“It’s still pretty stunning to me that simply by changing the software in the cell, the cell immediately starts this process of converting into another species,” J. Craig Venter says. “It’s all about how life works, how dynamic it is.”
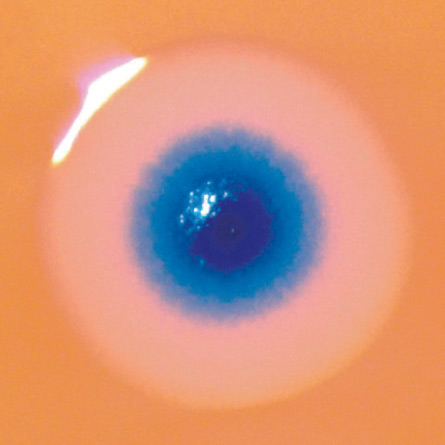
In most ways, the man-made genome was similar to the natural one, with a few important tweaks: The scientists added DNA sequences that the genome needs to survive the yeast-based assembly step. The team also added sequences encoding a substance that causes a cell to turn blue in the presence of certain drugs, making colonies of the synthetic bug identifiable with the naked eye. And finally, four unique genetic watermarks that can be used to unambiguously distinguish a synthetic M. mycoides cell from a naturally occurring one were included.
So far, Venter and his team haven’t engineered any special properties into the synthetic genome. “This is not so much about parts as it is a chassis to put the parts into,” says technologist George Church of Harvard Medical School in Boston.
Researchers have been tinkering with genes for many years, but this newfound ability to replace an entire genome is different, Venter says. Other studies typically change a small number of genes isolated from bacteria, he says. “Now we start with information in the computer. We start with digital code and create new genetic code from four bottles of chemicals [the A’s, T’s, G’s and C’s that make up DNA]. I think that’s the biggest philosophical difference.”
To some, though, this man-made genome is not technically artificial. “It’s a great feat, but I wouldn’t call it an artificial organism,” Collins says. Synthetic, he contends, implies designed from scratch, not plagiarized from a natural genome. What’s more, the experiment required a recipient cell to provide the cytoplasm to hold the transplanted genome. “It’s small, but it’s an important quibble,” he says.
To claim the creation of synthetic life, asserts Glenn McGee of the Center for Practical Bioethics in Kansas City, Mo., the entire organism must be successfully produced from raw materials.
“The landmark achievement has yet to occur,” McGee says. “What they’ve done is they’ve successfully transplanted DNA from one thing to another without noticeably harming the operation of the old DNA, as best they understand it, from their definition of its function. When I put it that way, it’s a hell of a lot less significant.”
Semantics aside, the real challenge is going to be turning this technology into something useful. “Our plan definitely is to move to the next level and make more complex and useful organisms that will have a lot of benefits to society,” Gibson says.
Designing genomes and transplanting them into microorganisms could lead to special bugs that produce vaccines, other pharmaceutical compounds and biofuels, for instance. Scientists at the Venter Institute are already working with Exxon Mobil to create bugs that slurp up carbon dioxide and convert it into clean fuel. Other applications include designer organisms that could convert wastewater into drinking water and clean up hazardous chemical spills.
Such efforts will require an incredibly detailed knowledge of the biology of the organisms, something scientists currently lack, Collins points out. “At best, we have a rudimentary understanding of these functions,” he says.
It may be that making targeted changes to existing genomes will prove just as successful as making organisms from scratch. “If the stated goal is to make useful microorganisms for commercial purposes, there are alternatives already in use,” says Harvard’s Church. “It’s not absolutely clear that suddenly a lot of people are going to adopt this method…. Nevertheless, it’s a big milestone.”






