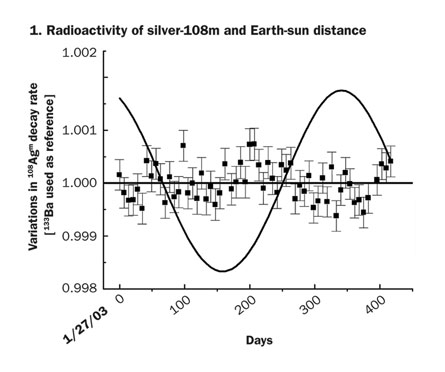
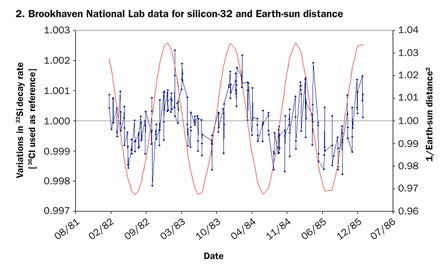
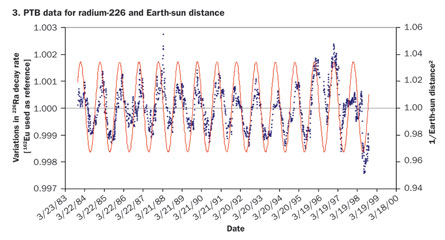
It’s nuclear physics 101: Radioactivity proceeds at its own pace. Each type of radioactive isotope, be it plutonium-238 or carbon-14, changes into another isotope or element at a specific, universal, immutable rate. This much has been known for more than a century, since Ernest Rutherford defined the notion of half-life—the time it takes for half of the atoms in a radioactive sample to transmute into something else. So when researchers suggested in August that the sun causes variations in the decay rates of isotopes of silicon, chlorine, radium and manganese, the physics community reacted with curiosity, but mostly with skepticism.
In one experiment, a team at Purdue University in West Lafayette, Ind., was monitoring a chunk of manganese-54 inside a radiation detector box to precisely measure the isotope’s half-life. At 9:37 p.m. on December 12, 2006, the instruments recorded a dip in radioactivity. At the same time, satellites on the day side of the Earth detected X-rays coming from the sun, signaling the beginning of a solar flare.
The sun’s atmosphere was spewing out matter, some of which would reach Earth the day after. Charged particles would contort the planet’s magnetic field, disrupt satellite communications and pose a threat to astronauts on the International Space Station.
But that dip in the manganese-54 radioactivity was not a coincidental experimental fluke, nor was it the solar flare discombobulating the measurements, the Purdue researchers claim in a paper posted online (arxiv.org/abs/0808.3156). In West Lafayette the sun had set while X-rays were hitting the atmosphere on the other side of the globe, and the electrically charged matter that created electromagnetic disturbances worldwide was still in transit. After a solar flare has begun, “the charged particles arrive several hours later,” points out theorist Ephraim Fischbach, coauthor of the paper with his Purdue colleague Jere Jenkins.
In a separate paper, also posted online in August, Fischbach, Jenkins and their collaborators compared puzzling and still unexplained results from two separate experiments from the 1980s—one on silicon-32 at the Brookhaven National Laboratory in Upton, N.Y., and the other on radium-226 done at the PTB, an institute that sets measurement standards for the German federal government. Both experiments had lasted several years, and both had seen seasonal variations of a few tenths of a percent in the decay rates of the respective isotopes.
A change of less than a percent may not sound like a lot. But if the change is real, rather than an anomaly in the detector, it would challenge the entire concept of half-life and even force physicists to rewrite their nuclear physics textbooks.
In those experiments, the decay rate changes may have been related to Earth’s orbit around the sun, the Purdue team says. In the Northern Hemisphere, Earth is closer to the sun in the winter than in the summer. So the sun may have been affecting the rate of decay, possibly through some physical mechanism that had never before been observed.
For example, the researchers say, the sun constantly emits neutrinos, subatomic particles produced in the nuclear reactions that power the sun. Neutrinos can move through the entire planet without being stopped, so the sun could affect radioactivity day and night. The closer to the sun, the denser the shower of neutrinos. Or the sun may emit fewer neutrinos during a solar flare, which would explain the December 2006 event.
Most physicists are dubious. For one thing, neutrinos interact negligibly with matter, so it’s not clear how they would affect radioactivity.
But some physicists take the results seriously and are searching old data for previously unnoticed effects. If the variations turn out to be genuine, theories may need revision, or new theories may be needed. “There’s no known theory that will predict something like this,” says theoretical physicist Rabindra Mohapatra of the University of Maryland in College Park.
If the results are confirmed, and nuclear decay is not immutable, perhaps physicists could find a way to speed it up to help get rid of waste from nuclear power plants. Such results might revise models of what goes on in the sun or change understanding of phenomena such as supernovas. Since neutrinos travel much faster than dangerous charged particles, using radioactive samples to detect solar flares when they first begin could prevent damage to satellites—and perhaps even save lives of astronauts.
Get a half-life
Some atomic nuclei are unstable, either because they are too big or they don’t have the right balance of protons and neutrons. Unstable nuclei decay by releasing different kinds of radiation, including energetic subatomic particles. For example, in beta radiation an excess neutron turns into a proton and spews out an electron—a beta particle—and an antineutrino. With an additional proton, the nucleus transmutes into a different element.
If a nuclide—a particular isotope of a given element—has a half-life of, say, one year, then after one year there will be half of it left. All atoms of a given nuclide are identical, and a one-year half-life means that each nucleus has a 50 percent chance of decaying over one year. If it doesn’t decay this year, it won’t be any more likely to decay next year—the odds will still be 50-50.
Half-lives are universal constants, as any physics textbook can attest. “Since Rutherford we’ve taken it as [a given] that decay rates are the way they are and nothing can change them,” Jenkins says.
Researchers use radioactive materials in a wide variety of applications where it’s useful to know the half-life with decent precision—the classic example being carbon-14, used in carbon dating of fossils. Usually, the half-life of a nuclide is measured in experiments that last just days or weeks. But for certain nuclides longer measurements are needed.
Between 1982 and 1986, a team led by David Alburger of Brookhaven monitored the radioactivity of silicon-32. The isotope’s half-life was known to be at least 60 years, so researchers needed a long time to measure it with any precision.
At the same time, the team monitored a chlorine-36 sample. Chlorine-36 has a half-life of more than 300,000 years, so a sample’s radioactivity stays virtually unchanged for a long time and can be used to spot any spurious fluctuations. To their surprise, the researchers found that both samples had rates of decay that varied with the seasons, by about 0.3 percent.
The samples were kept at constant temperature and humidity, so the changing seasons should have had no effect on the experiment. The team tried all the fixes it could to get rid of the fluctuations, but, in the end, decided to publish the results. No other lab tried to repeat the experiment, and the anomaly remained unexplained. “People just sort of forgot about it, I guess,” says Alburger, who retired shortly after the results came out.
Unbeknownst to Alburger, researchers at PTB in Germany had also found yearly oscillations in a decay rate, in a 15-year experiment on radium-226. (Two of those years overlapped with the Brookhaven experiment.) Now Fischbach and his collaborators’ comparison shows that the oscillations are in sync. Well, almost: Mysteriously, the peaks and troughs of the two oscillations seem shifted with respect to each other, by about a month.
Alburger says that the correlation between the patterns seen in his team’s data and the PTB’s is very convincing. “What causes it is the real question,” one that nuclear physicists should now look into, he says.
Mohapatra agrees that the effect looks genuine. But, he warns, genuine-looking effects are often later revealed as statistical flukes or the result of subtle defects in measuring technique. Still, he adds, “it’s interesting enough that people in the nuclear field should go back and look at old data.”
Take two
Peter Cooper of the Fermi National Accelerator Laboratory in Batavia, Ill., recently did just that. He obtained and analyzed data from the Cassini mission to Saturn. Deep-space probes usually generate power from the heat emitted by a chunk of radioactive material—plutonium-238 for the Cassini spacecraft. Cassini journeyed as close to the sun as Venus and then far back to Saturn, spanning a much wider range of distances from the sun than Earth does during its yearly orbit. If the sun had an effect on plutonium decay, the fluctuations would have been much more substantial than those seen in Earth-bound experiments. As a result, Cooper reasoned, Cassini should have measured substantial changes in its generator’s output. It didn’t. (His paper is posted online at arxiv.org/abs/0809.4248.)
Meanwhile, Eric Norman of the Lawrence Berkeley National Laboratory in California reanalyzed data from experiments on radioactive americium, barium, silver, titanium and tin, and found no seasonal variations, he says.
Fischbach is unfazed. Each nuclide, he notes, requires a different amount of energy to be nudged into decaying, and that the type of decay—be it alpha, beta or gamma radiation—may also play a role. “It’s possible that plutonium is inherently less sensitive than radium,” he says.
More recently, Fischbach found what he says is more evidence for his case. Exhibit A: An experiment on tritium, a radioactive isotope of hydrogen, which his collaborators are running at Purdue, may be measuring a seasonal effect, he says. Exhibit B: A 1990 paper by Kenneth Ellis of Baylor College of Medicine in Houston reported seasonal variations in plutonium-238 radioactivity in a calibration experiment for a radiotherapy machine.
But Fischbach, Jenkins and their colleagues have a lot of convincing to do, says Hamish Robertson of the University of Washington in Seattle. “There’s no physical basis for the decay rates to vary with anything, let alone with the Earth-sun distance,” he says.
Neutrinos in particular seem a very unlikely explanation to most physicists. Neutrinos only interact via the weak nuclear force, which has very short range, points out Boris Kayser, a neutrino theorist at Fermilab. And ordinary matter is mostly empty space. So detecting neutrinos is notoriously hard, Kayser explains. “Unless the detector is very big, so that it gives the neutrino many chances to come close to one of its particles, the neutrino will just go sailing right through it.”
Fischbach, though, says that perhaps neutrinos have a small electromagnetic interaction. While they have no electric charge, neutrinos carry a magnetic field. Instead of one neutrino giving a rare kick to one nucleus, a single neutrino could be giving “a small electromagnetic kick to a lot of nuclei,” potentially tipping the unstable ones into decaying. Fischbach admits that he hasn’t finished calculations to show that this would be possible.
The Purdue scientists are planning more experiments. In the end, the burden of proof will be on them, Cooper says. “Every experimentalist knows that the apparatus, or at least your understanding of it, is always at fault until demonstrated otherwise,” he says. It’s likely that seasonal weather caused the anomalies, he says, but admits that future work could prove him wrong. “Nature is really unmoved by what I, or anyone else, believes.”