Head Agony
Jumpy cells may underlie migraine’s sensory storm
When Lewis Carroll sent Alice down the rabbit hole, she encountered a strange and twisted land with distortions of size and time. Some headache experts see something else — the possible ghosts of the author’s migraines, which can leave victims temporarily blinded, nauseated, hallucinatory, numb, unable to concentrate or seeking shelter from painful stings of light and sound.

People with migraines travel between two worlds: one in which they are having a migraine and one in which they are not. “I’m very brave generally,” Tweedledum tells Alice, “only today I happen to have a headache.” But even after the headache is gone, migraine sufferers live with the dread of its return.
For more than a century, researchers have been trying to step through the looking glass to find clues to the mystery of migraines, with little success. Treatments that can prevent or end migraine attacks exist only because drugs for something else were found, often by accident, to quiet the migraine’s neurological storm.
“All of the major things we use were not designed for migraine at all,” says Peter Goadsby, a neurologist at the University of California, San Francisco. “It’s not good enough that one of the commonest of medical problems has treatment developed by serendipity.”
A major barrier to relief, it turns out, has been that migraines, which affect 36 million people in the United States, have no known cause. But researchers now think that they are, at least, looking for the culprits in the right places.
“For almost 100 years, people believed it was a vascular disease,” says Rami Burstein, academic director of the Comprehensive Headache Center at Beth Israel Deaconess Medical Center in Boston. Blood vessels, it is now thought, are probably just aiding and abetting the crime. Instead, the perpetrators are nerves inside the brain itself, which misfire and set off a disastrous sequence of events. Making things more complicated, migraine sufferers may differ in their thresholds of susceptibility and sets of triggers. “Migraine is for the most part a genetic disease,” Burstein says, “a problem with genes that regulate excitability of neurons in the cortex.”
Study results announced last year point to three genes that may be linked to migraines. Along with other “migraine genes,” the findings seem to paint a picture of brain cells that remain too jumpy when they are supposed to be settled down. With each new discovery, migraine sufferers are brought closer to the day when they might finally be able to escape the hell in their heads.
Stemming the tide
Migraine researchers spent a long time focusing on blood vessels, partly because the notion seemed to make sense: The pain of a migraine throbs in sync with a person’s heartbeat, and triptans — the first class of drugs approved specifically for migraines — constrict blood vessels in the brain. Research suggested that vessels in the head set off a domino of neurological reactions after abnormally contracting, then dilating. Ironically, some of the shift away from the blood vessel theory arose from the study of triptans themselves.
Sumatriptan, the first of the triptans, was introduced for clinical use in the early 1990s. “It was a real milestone in the headache world,” Goadsby says. “But as people looked at sumatriptan, it became clear that it did more than just constrict blood vessels.” The drug also affected nerves, raising the possibility of a different explanation for its effectiveness. More recent evidence has poked holes directly in the blood vessel theory: A study in 2008 in the journal Brain found that blood vessel size during much of the headache is normal (though a more recent study with better resolution is more supportive of at least some vascular role in a migraine attack).
Goadsby was drawn to study migraines largely because one part of the vascular theory never made sense to him. The pain often strikes only one side of the head, and it isn’t necessarily the same side from episode to episode. “I never understood how something originating from the bloodstream would just affect one side of the head,” he said.
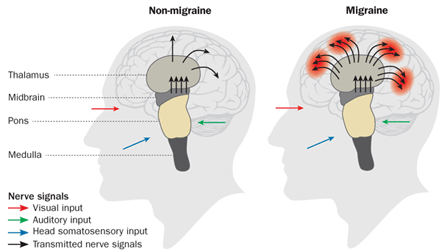
So Goadsby decided to use medical imaging as a window into the brains of people having a migraine attack. His curiosity had mostly to do with the brain stem, which controls many basic body functions, such as digestion and circulation — some of the same systems known to go haywire during a migraine attack. It seemed logical to him that a migraine had to originate in some central area since, other than the headache itself, the symptoms don’t tend to be lopsided.
“The brain stem has, in a small area, the ability to affect a large part of the brain,” Goadsby says. In experiments he began publishing in 2005, he used PET scans to identify three locations in the brain stem (corresponding to three distinct structures) that show greater than normal activity during a migraine attack. These parts of the brain usually act as a damper on nerve signals, he says. But when the structures allow too much input to pass through, nerve signals pour into the rest of the brain without control, Goadsby hypothesizes. That’s why normal sensations such as light, sound and even the pulsing of blood through the head become painful. He also believes that the malfunctioning of these areas of the brain stem give rise to one of the most peculiar aspects of a migraine — the aura.
An aura is a sensory disturbance that occurs in about one in four people with migraines. It commonly begins as a flickering of bright lights, like the flashes of an ambulance, in the corner of the visual field and gradually intensifies as it works its way across the line of sight. Some patients experience tingling, nausea and slurred speech during the aura, as well as blind spots once it subsides. An aura vanishes within an hour, and is replaced by a detonation of pain in the head.
Unbearable lightness
The origin of the aura has long been a topic of research fascination. An explanation now gaining consensus emerged in the 1940s from Brazilian biologist Aristides Leão while he worked on his dissertation at Harvard Medical School. After performing experiments in rabbits, Leão noted a marked change in electrical activity that started in one group of cells and rippled across the cortex, the outer layer of the brain. The hyper-excited nerves fire, and then appear to recalibrate, going temporarily silent. Leão called the phenomenon “cortical spreading depression.” Later researchers connected the phenomenon to a migraine aura, reasoning that the flickering light that patients see could occur as the cortical spreading depression crosses the visual center at the back of the brain.
The theory of cortical spreading depression was mostly confined to animal data, remaining at the margins of medical research for decades, until researchers could obtain brain scans of patients experiencing a migraine. One of the first of these images, published in 1994 in the New England Journal of Medicine, came about by chance. A young woman was in a scanner at UCLA for a study of cerebral blood flow when suddenly her migraine began. For about 15 minutes, researchers were able to document a wave of low blood flow (which they believe corresponded to nerve cells going temporarily inactive) spreading across the woman’s brain. A second study was led by Michael Moskowitz of Harvard Medical School. Moskowitz says that in the late 1990s he learned of an engineer who could induce his migraine auras with intense exercise. So Moskowitz and his team invited the man to campus, had him play basketball for 80 minutes and then whisked him off for an MRI.
In 2001 in the Proceedings of the Na-tional Academy of Sciences, Moskowitz’s team published the first MRIs of an aura from start to finish. In addition to the engineer, the researchers also took brain images of two campus employees who entered the scanner shortly after their auras had begun. As predicted, the images showed a wave of overactive (then underactive) neurons creeping across the cortex. “It’s not unlike what would happen if a stone were dropped in quiet pond,” Moskowitz says. The data suggest that the blood flow changes in the brain — the ones that led researchers in the wrong direction for decades — are more likely caused by the aura, and not vice versa. Active neurons grow hungry for fuel.
There is little debate about whether cortical spreading depression is a component of a migraine, but one central question is where it comes from, as well as its exact relationship with the pain. Goadsby believes the aura and pain arise from the brain stem, but others, like Moskowitz, say the activity changes observed in the brain stem are a result of a migraine, not the cause.
“The evidence that this kind of an event is triggered in any precise region of the brain is overstated,” Moskowitz says. Instead of having origins in the brain stem, he suggests, the cortical spreading depression can arise in any part of the brain, an idea consistent with animal studies.
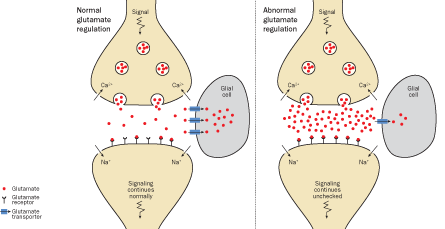
Moskowitz also does not agree that the changes that occur in the brain stem during a migraine open the floodgates on nerve signals, allowing normal sensations to become painful. Instead, he points to laboratory experiments supporting the idea that the cortical spreading depression itself is responsible for the pain. Most of the brain does not have the capacity to feel pain; only the nerve endings in the dura mater, the organ’s tough outer covering, and related tissue are pain-sensitive. A headache happens because of the stimulation of these nerves, which fire as the wave of excitability makes its way across the cortex, Moskowitz believes. “Cortical spreading depression releases in a drastic way a group of chemicals that are normally sequestered in cells of the brain,” he says. When those chemicals — such as potassium, hydrogen ions and the neurotransmitter glutamate — get dumped, they may activate the pain fibers on the brain’s surface through a process that is still being worked out.
Skeptics of this idea point out that most people get migraines without an aura. Moskowitz’s belief is that cortical spreading depression occurs but does not affect the brain’s visual processing and thus remains undetected — a kind of hidden aura.
In truth, much about migraines is still unknown, says Burstein of Beth Israel. “Migraine is a complex disorder,” he says, involving abnormalities in both the cortex and the brain stem. Where the pain comes from is still unclear, and perhaps the mechanisms behind chronic migraines (which occur more than 15 days a month) are different from those for less frequent ones. Migraines that strike regularly could originate from the cortex but over time could damage the cells of the brain stem. “This damage may reduce the ability of the brain stem to mitigate incoming signals,” he says.
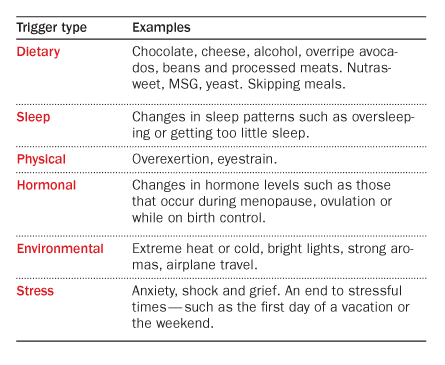
And none of the research so far has clearly answered the biggest question: What’s the first thing that goes wrong? The list of potential migraine triggers reads like a catalog of daily life experiences: hunger, flashing lights, alcohol, strong smells, chocolate, cheese, too little sleep, too much sleep, stress, relaxation after stress, menstrual cycle changes, weather changes, and on and on. (And there may be no triggers at all; the changes in the brain that lead to an attack could cause someone to crave chocolate or feel stressed, or undergo other physiological shifts.)
“We don’t know why someone gets a migraine when they are exposed to a perfume in the elevator, when 10 other days they smelled the perfume and didn’t get it,” Burstein says. “Once it starts, we know a lot about it. What we don’t know is why and how.”
Tinderbox of nerves
While scientists debate the why and how of the trigger, genetic studies are getting at a separate question — who. Most researchers believe that genes contribute to a person’s susceptibility to the triggers. The headaches seem to run in families, and the risk is higher if you have an immediate family member with migraines.
Most migraine sufferers probably possess a constellation of genes, each one of which contributes a little to the susceptibility. But there is one kind of migraine, called familial hemiplegic migraine, that appears to have an even stronger genetic component.
An international team of researchers studying this kind of migraine described the first “migraine gene” in the journal Cell in 1996. The discovery came after neurologist Michel Ferrari of Leiden University Medical Center in the Netherlands happened to see two patients on the same day with familial hemiplegic migraine. (They turned out to be both from the same region of the country and were obliquely related.)
Since then, researchers have discovered a handful of genes that may contribute. By understanding the role of these genes, scientists could gain clues to the mechanism behind a migraine. So far, all signs point to nerve activation.
“They confirm that migraine is a disease of hyperexcitability in the brain,” says Ferrari.
Nerve cells work by transmitting electrical impulses. Normally, the inside of a nerve cell is negatively charged (thanks to a lot of negatively charged chlorine ions) and the outside is positive (from positively charged sodium, calcium and potassium ions). When a nerve cell releases a signaling molecule called a neurotransmitter, channels on the cell next door open up and allow positive ions to rush in. The cell briefly depolarizes — the inside loses its negative charge — and then returns back to its normal state.
Genes that have been linked to migraine all have some role in the firing of nerve cells and this positive-negative ion swap. The gene mutation described in 1996 affected the calcium ion channel. Another DNA variation, described in 2010 in Nature Genetics, inhibits a cell’s ability to clear away the neurotransmitter glutamate after the nerve has fired, allowing it to accumulate. In June 2011, Ferrari and an international team of researchers described signs of three more rogue genes in Nature Genetics. These, too, are involved in the transmission of signals from cell to cell.
“The story seems to go in the same direction,” Ferrari says. In a migraine-susceptible brain “it’s easier to trigger neuronal activity.”
The idea is also supported by a study published last January in the journal Neurology. Researchers exposed patients suffering a migraine attack to light — a normal stimulus that becomes excruciating during the headache. PET scans of the patients revealed that the light caused the nerves in the occipital cortex of the brain to fire. But when the patients were not experiencing an attack, the light did not have an effect on that part of the brain. Like a drought-stricken forest, the nerves may be easy to ignite, and easy to fuel once they do.
“The fact of the matter is there is plenty of evidence now that the brain of a migraine sufferer is never normal,” says David Dodick, a neurologist at the Mayo Clinic in Scottsdale, Ariz., and president of the American Headache Society. It now appears that a migraine brain exists on edge, quick to set off a headache when the right combination of circumstances comes along. “Networks are active when normally they shouldn’t be,” Dodick says. “The threshold for generating an attack is always just below the surface.” The rabbit hole is always near.
But maybe not forever. Soon migraine sufferers may, like Alice, be able to wake up from the nightmare inside their heads — something Alice’s creator could never do.