Mom’s weight during pregnancy shapes baby’s health
Recent studies link maternal obesity to child’s risk of mental and physical problems

MATERNAL INPUT Obesity at conception or during pregnancy is a big problem that's getting bigger: New evidence says a child's mental health could be at stake.
adrian825/iStockphoto
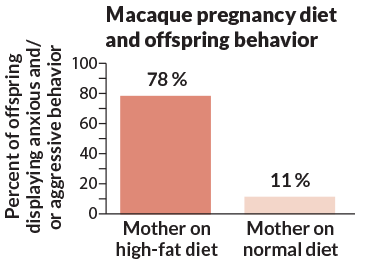
When Elinor Sullivan was a postdoctoral fellow at Oregon Health & Science University in Portland, she set out to explore the influence of food and exercise habits on obesity. In one experiment, she and her colleagues fed a troop of macaque monkeys regular chow. Other macaques dined American-style, with a hefty 32 percent of calories from fat and ready access to peanut butter treats. Over time, the second group of monkeys grew noticeably fatter.
Then they all had babies.
Sullivan, now at the University of Portland, noticed odd behavior in the plump moms’ offspring. At playtime, they often slinked off by themselves. When handled by keepers, the infants tended to vocalize anxiously, and the males became aggressive. They were prone to repetitive habits, like pacing.
In their carefully controlled world, the only difference between those monkeys and others at the facility was their mothers’ extra pounds and indulgent diet. The behavior was so striking that Sullivan changed the course of her research.
“It made me start thinking about human children,” she says, and the twin epidemics of obesity and behavioral problems such as attention-deficit/hyperactivity disorder. Her research, published in 2010 in the Journal of Neuroscience, was one of the first studies to note that the progeny of female monkeys eating a high-fat diet were more likely to experience altered brain development and suffer anxiety. Not long after, researchers worldwide began compiling evidence linking the heaviness of human mothers to mental health in their children. One headline-grabbing study of more than 1,000 births, reported in 2012, found that autism spectrum disorders showed up more often in children of obese mothers than in normal-weight women (SN: 5/19/12, p. 16).
Over the course of a generation, obesity rates among U.S. women have soared. Today, 38 percent of females in the population are obese (defined as a body mass index of 30 or higher). Among women of childbearing age, well over half are overweight or obese, with almost 8 percent considered extremely obese (a BMI of 40 or greater). Lucilla Poston, who is head of the division of women’s health at King’s College London, calls too much weight during pregnancy “the biggest problem in obstetrics at the moment.”
Within the body, obesity is not a passive state. Excess weight can inflame the immune system, upset the balance of hormones and even alter the microbial flora tucked inside the intestine. If shared by the fetus, any or all of these changes can affect the baby’s development in subtle but important ways. Further complicating matters, the fetus is probably being exposed to the effects of fattening, and perhaps inflammatory, foods.
Only recently have researchers begun to understand what this physiological storm might mean for children. In part, obesity during pregnancy raises the odds that a baby will be born overly large, setting the stage for future health problems (SN: 5/31/14, p. 22). But when a mother is excessively overweight, risks persist even for newborns of normal size. One study published in 2013 in the journal BMJ analyzed medical records of more than 37,000 people born in Scotland between 1950 and 1976. After accounting for socioeconomic status, gender, weight at birth and many other variables, the researchers found that children born to obese mothers had a 35 percent higher mortality rate from birth to 2012. “Independent of birth weight, a child can grow up with increased blood pressure, obesity and risk of diabetes,” Poston says.
The list doesn’t stop there. Perhaps most surprisingly, a mom’s metabolic state might compromise her child’s mental health—the very observation that changed Elinor Sullivan’s career. One study published in 2015 even raises the possibility that a child’s normal cognitive development might be slightly impaired by mom’s high BMI.
If there is a bright spot, it’s that unlike many threats during development, this one is preventable. As the risks of obesity during pregnancy emerge, researchers hope more young women on the verge of starting families see the importance of maintaining a healthy life—and that the culture around them will support efforts to do so. “Pregnancy is a good time to talk to people about lifestyle,” Poston says, “because they do care deeply about their babies.”
Womb with a clue
Few research questions are easy, but epidemiologists studying maternal obesity face a particularly daunting challenge. They have to separate the effects of a mother’s weight from a multitude of other influences on children’s health. In the United States, obesity disproportionately affects low income and minority women. Children born in less affluent neighborhoods face obstacles to their well-being: more stress, heightened exposure to pollutants and less access to wholesome foods. Plus, the same food choices and lack of activity that drive a woman’s weight gain may also become the lifestyle adopted by her children.
The data become even more difficult to tease apart when examining effects on the mind. Given the correlation of obesity with poverty, children of obese parents also might have educational disadvantages. Case in point: Studies have found that young children in poverty score lower on measures of school readiness, including motor-skill development, emotional health and social knowledge.
That said, the latest studies—many published in recent months—attempt to overcome those biases. And they still find reason for concern. Lisa Bodnar, a nutritional epidemiologist at the University of Pittsburgh, describes a “small but growing literature” suggesting that obesity in a mother is associated with lower cognition and other mental health challenges in children. In 2015 in the Journal of Nutrition, Bodnar and her colleagues published a study of women on similar economic footing who were patients at Pittsburgh’s Magee Women’s Hospital. The majority were unemployed, single mothers. The researchers nonetheless found that children of women who were obese at conception or gained excess weight during pregnancy scored slightly lower on tests of intelligence and executive function, a measure of the ability to plan, organize and adjust to new situations.
Probably the most compelling data link maternal obesity with ADHD, says Sullivan, who continues her primate studies. Whether maternal obesity (or a fattening diet) can actually cause hyperactivity is unclear, but one study of rodents published in Molecular Psychiatry in 2012 described results that “point to a direct biological link between in utero exposure to maternal obesity and hyperactivity in the adult offspring.” Researchers from England and Sweden fed one group of female mice a high-fat diet that started six months before pregnancy and lasted until weaning, while another group ate regularly. The offspring of the obese mothers scored significantly higher on tests of hyperactivity.
Another animal study, published in 2014 in the Journal of Neuroinflammation, found that female offspring of mice fed a high-fat diet had increased anxiety while the males were prone to hyperactivity. The study, from the Mayo Clinic in Rochester, Minn., and Oregon Health & Science University, also opened the door to prevention. When mother rats were given a healthier, less inflammatory diet while nursing, the mental health of the female pups improved, though the males still had issues.
In November 2015, Sullivan and colleagues reviewed the evidence in Hormones and Behavior, making the grim prediction that, given persistent rates of obesity and pervasiveness of high-calorie foods, “the prevalence of neurodevelopmental and mental health disorders will continue to rise in future generations.” In December, researchers from George Washington University and Mathematica Policy Research announced that 12 percent of U.S. children and adolescents have been diagnosed with ADHD, a 43 percent increase since 2003.
The field is still too new to explain biologically how obesity would impair fetal brain development, but Sullivan points to theoretical consequences of high glucose or the hormone leptin. Leptin inhibits appetite, but is often elevated in obese individuals and may affect brain development. Most commonly, however, researchers circle back to the effects of an agitated immune system on the brain. “We think of obesity as the state of chronic inflammation,” Sullivan says. “Many of the neurotransmitters in the brain are very sensitive [to inflammation] in early development.”
Baby buggy
The immune system isn’t the only part of body mechanics co-opted by obesity and diet. A compelling line of inquiry has linked the microbiome—specifically the microorganisms inside the digestive system— to body weight. For example, the microbiome of an obese person differs from the microbiome of someone of normal weight. In experiments involving lean mice with no intestinal microbes, transferring the microbiome of an obese person to a thin mouse is enough by itself to make the lean mouse pack on weight.
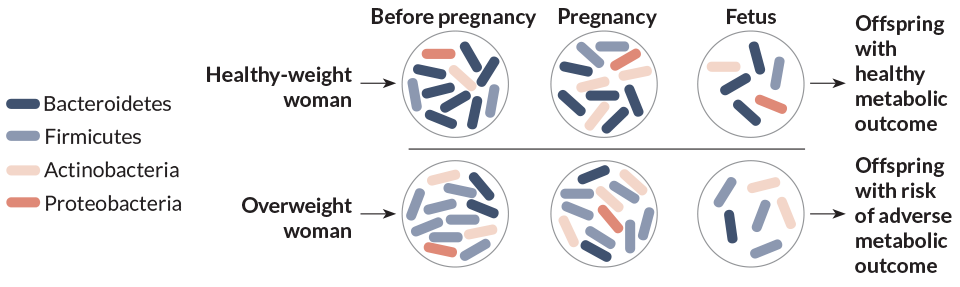
Since a newborn gets its microbiome from mom, a baby could inherit microbes that want to hoard calories. In both human and animal studies, the microbiomes of offspring born to obese moms are different than in children born to lean moms, says Deborah Sloboda, a fetal physiologist at McMaster University in Hamilton, Ontario. “What we don’t know is whether it comes from transfer during pregnancy, transfer during birth or poor developmental environment altering how the gut forms.”
The intestine is normally a fortress that does not like microorganisms to escape. Several studies, however, have suggested that when under siege from a fast-food Western diet, the gut lining can become porous (SN: 5/30/15, p. 18). Perhaps bacteria slipping into the bloodstream during pregnancy could affect proper formation of the intestine. Other scenarios are also possible: The microbiome transferred during birth could, as it has in animal experiments, predispose a child to a microbiome that extracts more calories from a given amount of food.
In Pediatric Research in 2015, Sloboda and colleagues reviewed research on obesity and the maternal microbiome. One theory holds, they noted,that the microbiomes of lean women remain stable during pregnancy; the microbiomes of obese women appear more volatile, experiencing a greater bloom of species associated with obesity. These women’s children may then start life with a microbiome inclined toward weight gain.
Story continues after graphic
Provoking problems
Like researchers who study the brain, Sloboda and others suspect that inflammation—which also appears to be a consequence of the microbiome coping with junk food—lies at the heart of many risks conveyed to a developing fetus. “When you consider the spectrum of conditions that have been linked to maternal obesity,” says immunologist Ilhem Messaoudi of the University of California, Riverside, “one of the things that links all these diseases is inflammation.”
In addition to the irritation that might come from the high-fat, high-sodium, high-calorie fare at the drive-through, adipose tissue itself provokes a mother’s immune system. In this state of overactivation, the normal cues for her baby’s immune formation might then become lost.
“If you have to develop an immune system in the presence of inflammation, the programming of the immune system is going to change,” Messaoudi says. In an experiment published in 2015 in Pediatric Allergy and Immunology, she and her colleagues studied 39 pregnant women who were designated as lean, overweight or obese, based on their preconception body mass index, a measurement of body fat. The researchers extracted blood samples from the umbilical cords of the women’s newborns, and tested the reaction to antigens, molecules that are supposed to trigger an immune reaction.
“The cord blood cells of babies born to obese moms did not respond to bacterial antigens,” she says. It was as if the immune system, put to its first real test, was stumped. “If your immune cells don’t know how to react, you’re going to be sick more often. You may not respond to vaccinations in the way your immune system is supposed to respond.”
Those findings may partially explain studies finding that children of obese mothers are more likely to develop disorders that arise from off-kilter immunity. In 2014, researchers who reviewed a dozen studies concluded in the journal Pediatrics that babies born to mothers with a high body mass index had a 20 to 30 percent greater risk of asthma and wheezing, though they noted that mechanisms remain unknown.
Rewired appetite
Of all the possible consequences of maternal obesity, the data are most compelling in suggesting that overweight mothers tend to raise children who grow up to be overweight themselves. “It’s a very strong effect, and consistent, across all populations,” says Bodnar, from Pittsburgh. Chinese researchers writing in 2013 in PLOS ONE pooled the analysis of 45 studies examining whether children faced greater odds of being heavy based on mom’s size. Although studies have varied and genetics obviously play some role, the scientists concluded that having an obese mother roughly tripled the risk of obesity.
In addition to a woman’s weight when she becomes pregnant, excessive weight gain during pregnancy, especially in the first months, is also linked to her child’s obesity risk. In one 2012 study comparing more than 6,600 Finnish mothers, those who put on more pounds during the first 20 weeks of gestation (compared with those who gained the least) had children who were 46 percent more likely to be overweight at age 16.
Theories to explain the association are examining how increased glucose and hormonal balance affect fetal development, particularly in the brain. Leptin resistance, which leads to higher secretion of the hormone, can be a consequence of obesity. In the journal Acta Physiologica in 2014, Poston and her colleagues from King’s College pointed out that many studies have found that the presence of too much leptin can cause collateral damage to the developing hypothalamus, a key interface between the brain and the hormone-producing endocrine system.
Animal studies suggest an altered hypothalamus could mean a child is born with difficulty regulating blood pressure and controlling appetite. “That particular part of the brain may become rewired, and a child may grow up eating more,” Poston says.
With little sign that the obesity epidemic is abating, that theory and others are likely to take on increasing importance in medical research. This generation’s greatest health threat could leave an unexpected legacy. Scientists working in this field often worry that their research will be seen as solely finding fault with mothers. “I think it’s unfair to blame this on women,” Bodnar says. Obesity is a global problem. One starting point, she says, is for more doctors to speak with their patients about the importance of weight. Since half of all pregnancies are unplanned, those conversations should happen before a woman gets pregnant. Yet in a U.S. study published in 2014, overweight women of child-bearing age received diet and exercise advice during preventive medicine exams only 36 percent of the time. The number was even lower among pregnant women who were overweight.
At the same time, Bodnar says, this is not going to be fixed in doctors’ offices. Women every day are offered cheap, calorie-dense food, pushed by companies with fat marketing budgets (McDonald’s alone spends about $900 million a year on advertising). “It’s not easy, in this environment, to lose weight,” Bodnar says. “We have to agree as a society that this matters.”
This article appears in the January 23, 2016, issue of Science News under the headline, “Maternal Input: A mother’s weight during pregnancy can shape her child’s mental and physical health.”