New rules for cellular entry may aid antibiotic development
Tests show clues to fighting drug-resistant gram-negative bacteria
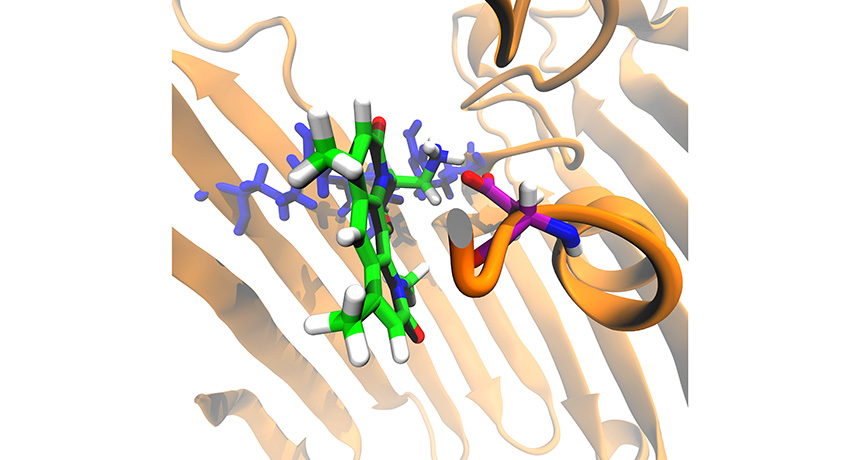
MAKE WAY An antimicrobial (mostly green and white in this illustration) that has been modified to target gram-negative bacteria travels through a channel (brown) on its way inside an E. coli cell.
M.F. Richter et al/Nature 2017