A partnership apart
DNA in hand, scientists dissect and redefine the iconic lichen mutualism
After spending an afternoon with François Lutzoni, it’s hard to understand why more sports teams aren’t named for lichens. Or why lovers bother with roses instead of sending a dozen fruticose lichen thalli. Lichens, Lutzoni explains, form when living organisms mingle intimately and become something more complex, capable and gorgeous than they could ever be alone.
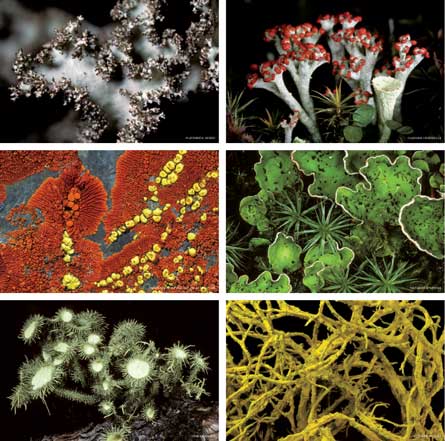

A long-time classic in discussions of taxonomically odd couples, lichens may form even more bizarre households than specialists had thought. Lutzoni’s lab at Duke University in Durham, N.C., among others, is using DNA analysis to shake up the old textbook truths about these partnerships.
To the extent that anyone remembers what biology books say about lichens, the memorized nugget probably has to do with a fungus entwining a photosynthesizing microbe. The green partner is a kind of alga or a cyanobacterium. In some cases, both join in.
One thing basic textbooks don’t mention, because researchers are only now exploring it, is the presence of other fungi lurking inside the supposed twosome or threesome. Every lichen species tested so far contains multitudes, says Lutzoni. Going into the field to sample lurker diversity leads to insane — his word —amounts of sample processing in makeshift labs (including, on occasion, bathrooms). Not to mention the discussions with airport security over the team’s giant suitcase packed with 3,000 tubes of lichen specks.
What those fungal lurkers do in lichens isn’t yet clear. But DNA sequencing should at least help Lutzoni and his collaborators detect these additional fungi. And that’s just one of many examples of how modern genetic techniques are being used to reveal new aspects of an age-old partnership. Biologists are learning how such an extreme cooperation evolved and what its history hints at for other symbioses. New studies are also illuminating how the parties meet and greet to set up their partnership. Or maybe that’s partnerships, plural. It may take a village to raise a lichen. Or Lutzoni and his team may discover a lichen Tokyo.
Duo dynamics
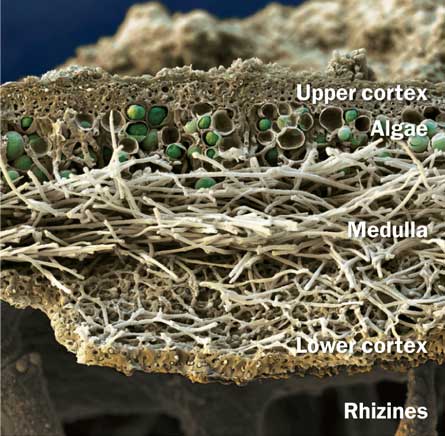
In the traditional lichen relationship, the alga or cyanobacterium provides sugar as food for the fungus, and the fungus provides other nutrients and a house — perhaps protecting its partner. Other creatures also form mutualisms, the term used for symbiotic living arrangements that benefit both partners. But lichens attract special attention because of the gap between the sum and the parts.
To describe the lichens that best exemplify that gap, Lutzoni and Duke colleague Jolanta Miadlikowska flip through the photographs in the tome Lichens of North America. Lutzoni taps a picture of lichens nicknamed British soldiers — ranks of stout, green columns wearing very red hats. Miadlikowska muses over the portrait of lichens in the shape of wine goblets, slim stems rising into perfectly round cups. More pages flip, and then, “Usnea!” Lutzoni points to what look like tiny escapees from Dr. Seuss’ drawing board.
The book also shows swaths of netting as airy as Halloween cobwebs, beards of lichen strands in Tour de France yellow, Jackson Pollock spatters in flame tones and pelts of overlapping forest-green lobes. The greenery belongs to a Peltigera, a lichen that has already won the team some cover space on scientific journals. “Got Peltigera?” says a bumper sticker on a lab door. Lichens definitely got diversity.
Lutzoni strides down a hallway and unlocks a door into a compact space thrumming with refrigerator noise. Here, the lab grows the symbiotic partners separately for genetic studies.
Lutzoni peers at racks of petri dishes filled with clouded or rust-colored disks of culture medium and plucks out a dish housing the algal half of a pixie cup lichen. The dish holds some green dots. “Here’s a better one,” he says. Instead of a dot, it has a streak. On another shelf, he finds the fungal partner, a faintly flesh-colored network about the size of a quarter. For fungal pizzazz, this culture plate loses out to most office fridges.
Fungi that form lichens often prove a headache to grow in the lab, Lutzoni says. He bristles at the textbook dogma that, in a lichen, the big, tough fungus protects the frail, photosynthesizing microbe. Googling “lichen fungi protect algae” turns up plenty of these slights to the green partner. Yet the challenges of growing enough of the fungi for DNA work reveals the fungi on their own as neither big nor tough.
In contrast, Lutzoni doesn’t find the Trebouxia alga he studies particularly frail. What researchers have discovered is that it does really well by itself, he says. Though Trebouxia algae don’t typically grow a lot on their own, they don’t shrivel up and die waiting for a fungus. “It’s great that the algae can be there in a dormant state waiting for Prince Charming.” This sturdiness makes sense in terms of lichen formation: Each time a fungus or alga reproduces, as each can when configured in a lichen, the next generations have to get together with new partners. Biologists have yet to report more than one loner lichenizing fungus thriving in nature.
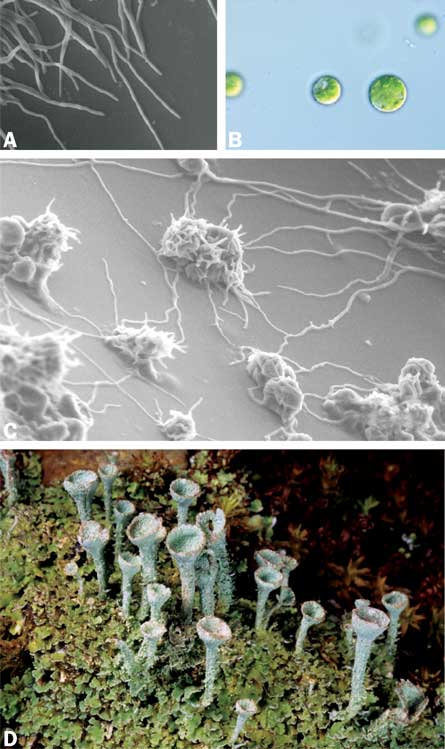
In the hallway of Lutzoni’s lab, a tablecloth-sized poster shows meet-ups between new partners. The first image shows a young, available fungus elongating as a thread. An algal cell, plump as a bath bead, sits near the thread’s path. In the next image, the two have met. Once it finds the alga, the fungus switches modes, its thread forking into a network of tendrils that embrace the alga.
Picky fungal farmers
Modern genetic techniques are giving researchers a new window into lichen development. Suzanne Joneson, now at the University of Idaho in Moscow, and her collaborators teased the potential symbionts, putting a membrane between the two so they could sense each other but couldn’t touch. Monitoring which genes surge in which partner at this phase, researchers see a lot of fungal gene activity but less by the alga.
But fungi don’t snuggle up with just anything. In the summer issue of Symbiosis, Joneson and Lutzoni describe other experiments in the fungal singles scene. When Joneson and Lutzoni offered the fungus Cladonia grayi a range of possible partners, including teensy glass beads, only some of the pairings triggered the abundant branching typical of the first stage of lichen making. Biologists have speculated that much of lichen initiation is driven by thigmotropism, or a tendency to grow toward physical contact. Turns out the contact alone is not enough.
As evident in these studies of lichen initiation and gene activity, the fungus seems the more aggressive, says lichenologist and ecologist Robert Lücking of the Field Museum in Chicago. He quotes a quip from a colleague: “Lichens are just fungi that have discovered agriculture.”
His imagery captures a considerable disparity that lichenologists have long noticed. From the intricate structures the combos can produce, showy wine goblets or infantry, a nonlichenologist might expect that the partners coevolved over millennia to form close pairs, with each fungus committed to only one alga. Believe it or not, that’s wrong too, says Lücking. At the current tally, more than 17,000 species of fungi form lichens in partnership with a total of only a few hundred species of algae or cyanobacteria. Modern genetic analysis is refining species counts, but Lücking predicts that once the DNA settles, the huge disparity will remain. Instead of Fred Astaire and Ginger Rogers, lichens are more like specialty farmers and their crops. Several sell heirloom tomatoes while others tend Gala apples.
Lücking takes the farming idea a step further with a particularly tidy new example in the August issue of the American Journal of Botany. In sequencing samples of one of the big genera of lichen cyanobacteria, up until now all classified as Scytonema, the researchers found what they propose calling a new genus. These lichen-forming cyanobacteria all belong to a lineage quite different from nonlichenized Scytonema. It’s as if fungi, in effect, domesticated a crop that’s as distinct from its ancestors as barbecued sweet corn is from its wild ancestor, the skinny, scrubby teosinte.
By convention, the cyanobacterium and alga in a lichen don’t get the billing of Black Krim tomatoes grown by specialty farmers. Lichenologists use the Latin name of the fungus to double as the name for the partnership. Lutzoni says that, no, he doesn’t smolder with outrage for underacknowledged photosynthesizers. “It’s a slippery slope,” he says. Leaps forward in understanding symbiotic mutualisms have revealed so many others — in bioluminescent squid organs and aphid guts and plant roots — that giving a separate name to one symbiotic mutualism might set off a taxonomic tsunami. “You’d have to give new names to trees,” he says. “And we have gut bacteria.”
Fungi in trees
The perils of renaming symbioses highlight just how big a deal they are, which makes understanding the path of lichen evolution all the more interesting. Lichenizing fungi typically don’t have a lot of fancy morphology like flowers or bones that systematists can use for classification. So DNA sequencing is illuminating a lot of once-murky relationships.
Lichenizing fungi pop up on different branches of the fungi family tree. In the fungal phylum Ascomycota, eight to 11 of the orders (depending on the classification scheme) each mix lichen formers with nonlichenizing fungi. Lutzoni points out that two scenarios could have produced such a scattered pattern on the family tree. Either a lot of fungal lineages could have turned into lichenizers on their own, or a few way-back ancestors could have acquired a knack that a lot of their descendants have since lost. Debate continues on the details, but data so far suggest the second scenario.
A 1995 paper in Science reported a DNA-based family tree with five origins of the lichen habit, including two origins within Ascomycota. In 2001, Lutzoni and colleagues argued that lichens evolved earlier than thought within this phylum, possibly only once. A 2009 paper puts the number between four and seven.
One of the most interesting switches between regular fungi and lichenizers shows up among the relatives of Verrucariales lichenizer fungi, according to a 2008 paper in Studies in Mycology by an international team, including the Duke lab. In this new family tree, these lichenizers turn out to be closely related to black yeasts. There are few lichenizers among these yeasts, of the order Chaetothyriales, but the group does have some disagreeable pathogens that attack humans and other animals, including two that cause fatal brain infections.
The common ancestors of these two orders probably colonized bare rock, the researchers say. To Lutzoni, this family relationship suggests that an ability to cope with the extreme heat and drought in rocky places could have been a stepping stone to two symbioses, pathogenic activity in warm-blooded animals and lichen formation.
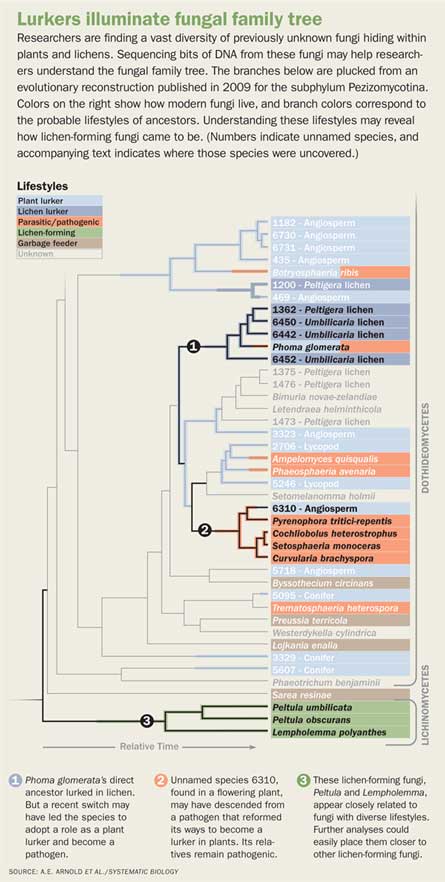
Ancestral lichens may have been especially important as the cradle for the huge number of fungi that biologists are finding lurking in plant leaves, Lutzoni, A. Elizabeth Arnold of the University of Arizona in Tucson and colleagues propose in the June Systematic Biology. All plant species that scientists have checked contain fungi growing inside the leaves, stems or other aboveground tissue. These fungi aren’t causing disease symptoms and may in fact help protect against disease, Arnold says. Leaves of wild cacao trees with abundant internal fungi, for example, put up especially strong resistance to colonization by a disease-causing fungus, she and her colleagues reported in 2003.
After working with cacao leaves, Arnold tried looking for extra fungal species lurking in lichens. She sterilized the surfaces of lichen snippets and set them in culture plates to see if threadlike fungal hyphae would grow from deep within. “We would watch these beautiful fuzzy, or feathery, or furry hyphal tips grow out,” she says. As far as colors on the culture plate, “you name it,” she says. “If you hold it up to the light, it’s like looking at a stained glass window.” And that’s how Arnold and Lutzoni became interested in the massive diversity of lurkers in lichens.
Many lurkers in these samples are new to science, and researchers still don’t know how lurkers make their living. Adding their DNA to a family tree of other fungal sequences is revealing how evolution has led to different fungal lifestyles. Tracing back the branch points in the family tree reveals spots where ancestral lineages switched among lichenizers, pathogens, lurkers and garbage feeders. The first glimmers of the pattern are consistent with Arnold’s idea that ancient lichens served as an incubator for lineages of fungal lurkers whose descendants have since switched things up.
Working through these ideas has led to the current effort to collect more samples and create a family tree with enough twigs to reveal patterns clearly. Lutzoni and Arnold are analyzing literally thousands of lichen samples from the dry, hot Arizona desert to the tundra of Alaska — explaining all the airline troubles. If the emerging lurkers turn out to be valuable partners in a multispecies complex, as they are in some plants, then lichenologists may have the pleasure of saying that lichens did it first.