Portrait of a cancer drug at work
As researchers seek better drugs for breast cancer, they have a revealing new picture that could guide their way. It shows where the breast cancer drug Herceptin binds to and inhibits the protein HER2 on cancer-cell surfaces.
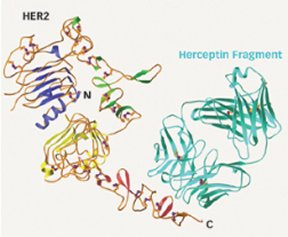
In 20 to 30 percent of breast cancer cases, overproduction of HER2 stimulates cell proliferation. To determine the structure of HER2 at the atomic level, Daniel J. Leahy, a Howard Hughes Medical Institute investigator at Johns Hopkins University School of Medicine in Baltimore and his colleagues shined X rays through crystallized HER2 with and without fragments of Herceptin bound to it. “It gives people a view of the exact interactions” between the drug and its target, says Leahy, whose team reports its work in the Feb. 13 Nature.
The newly revealed HER2 structure also explains why the protein is a powerful cancer promoter. Unlike related proteins, HER2 is normally in an active state and doesn’t need to be triggered by another molecule, says Leahy.
****************
If you have a comment on this article that you would like considered for publication in Science News, please send it to editors@sciencenews.org.







