Robert Lefkowitz of Duke University in Durham, N.C., and Brian Kobilka of Stanford University will share the 2012 Nobel Prize in chemistry for work on molecules that help cells communicate with the outside world.

The molecules, called G-protein–coupled receptors, sit in a cell’s outer membrane and identify hormones, odor molecules and other chemicals, transmitting that information to the inside of the cell.
“They are crucially positioned to regulate virtually every known physiological process in humans,” Lefkowitz said in a telephone call with the Nobel committee October 10.
Humans have hundreds of different G-protein–coupled receptors, about half of which detect odors. Others are involved in transmitting information from hormones or other chemical messengers. Some gather light in the eye, while others in the tongue sense tastes. Insulin, pain signals and the fight-or-flight reaction all depend on the receptors. Many drugs — by some estimates up to half of medications on the market — work through the receptors, including antihistamines, some psychiatric medicines, and beta-blocker drugs used to treat heart arrhythmias, high blood pressure and other cardiovascular problems.
“It’s like discovering the phone system in the United States if you didn’t know the phone system existed,” says Jack Dixon, the vice president and chief scientific officer of the Howard Hughes Medical Institute, which supports Lefkowitz’s work. Lefkowitz “has been a force in this field for more than 30 years,” and could legitimately be called its father, Dixon says. Kobilka is a newer player, but “they have both made seminal contributions.”
In particular, recent work by Kobilka establishes how the receptors act down to the atomic level and could fundamentally change how drugs are designed, says Kendall Blumer, a cell biologist at Washington University in St. Louis. “In the past we were doing this in the dark because there was no structural information available,” Blumer says. The new detailed picture of the receptors “will move [drug design] forward in ways that will really touch the lives of virtually every family.”
Lefkowitz and Kobilka’s Nobel-winning research began in 1968 with Lefkowitz’s efforts to understand how adrenaline works in the body. Using radioactively tagged adrenaline as bait, Lefkowitz fished for molecules on the cell’s surface that interact with the hormone and reeled in the beta-adrenergic receptor.
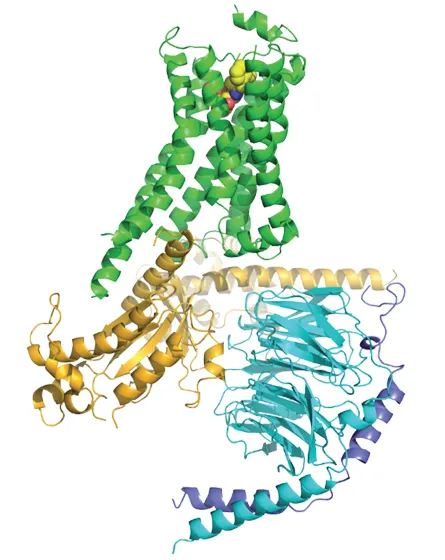
Follow-up research showed that the receptors function as part of a triumvirate, with adrenaline or other chemical messengers outside the cell and molecules called G-proteins inside. When a chemical such as adrenaline or insulin docks with a G-protein–coupled receptor in the cell’s membrane, the receptor, which resembles a bundle of sticks, changes its shape. That contortion tells the G-protein inside the cell to initiate a chain reaction, which then delivers the information to the rest of the cell.
Lefkowitz brought Kobilka onto his team in the 1980s. Kobilka’s job was to identify the gene that encodes the beta-adrenergic receptor protein. With the gene’s makeup in hand, the researchers could see that the protein corkscrews through a cell’s outer membrane seven times. That is the same configuration a light-gathering protein in the eye, called rhodopsin, adopts.
“I think that was the eureka moment,” Kobilka said of the realization that the two functionally distinct receptor proteins look very much alike. Since then, researchers have discovered that receptors that detect brain chemicals, odors, taste, some hormones, histamine and many other chemicals all snake through a cell’s membrane seven times.
In 2007, Kobilka’s group made the first picture of a G-protein–coupled receptor using a technique called X-ray crystallography. And just last year, Kobilka snapped a picture of a G-protein–coupled receptor in the act of transmitting a signal from a hormone outside the cell to a G-protein inside. It was an accomplishment years in the making, but showed Kobilka and his colleagues that transmitting messages involves some acrobatics on the part of the G-protein, an unexpected finding. The portrait also shows that subtle changes in the receptor’s shape in response to docking with different chemicals can lead to different messages being sent inside the cell, Blumer says.
Although he struggled for many years to get a 3-D picture of a receptor, Kobilka says all the hard work was worth it.
“Being a scientist is a lot of fun, even though it is delayed gratification and you fail a lot,” Kobilka said. He credits many collaborators over decades for helping decipher how the receptors monitor the outside world and tell the body how to react.
Lefkowitz and Kobilka will share prize money of 8 million Swedish kronor, about $1.2 million.







