Editor’s Note: Last summer, Science News posted a story about salt found in Saturn’s E-ring and its implications for a possible ocean inside Enceladus, the moon that supplies the particles for that ring. Now, the two studies cited in that story have both been published in the June 25 Nature. We’ve updated the story – the last three paragraphs – to include some additional interpretations of the findings.
LINK TO ORIGINAL POST Saturn’s moon may host an ocean
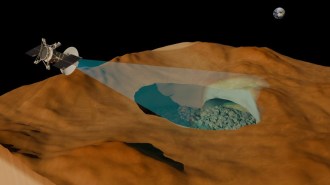
The Cassini spacecraft has found what may be the strongest evidence yet that Saturn’s tiny moon Enceladus has an ocean beneath its icy surface. If the liquid water finding is confirmed, it would suggest that the moon may be one of the most promising places in the solar system to search for signs of past or present extraterrestrial life.
Enceladus is already known to vent geysers of water-ice and vapor that contain complex organic compounds. The new evidence for an underground ocean comes from the detection of sodium in Saturn’s E ring, the extensive band of ice particles believed to be fed and replenished by Enceladus.
Cassini’s cosmic dust detector has recorded sodium in concentrations of about one part in 100,000 within the ring, Sascha Kempf of the Max Planck Institute for Nuclear Physics in Heidelberg, Germany, reported last month at a Cassini project science group meeting in Rome.
Although Kempf and his colleagues were initially concerned that some of the sodium might simply be a contaminant on their instrument’s detectors, his team is now confident that all the measured sodium is from the E ring, Kempf told Science News.
An ocean beneath the surface of Enceladus is the best way to account for the sodium, says Jonathan Lunine of the University of Arizona in Tucson, a Cassini researcher not part of Kempf’s team. Sodium resides in rock, he notes. It has been detected in regions of the solar system where charged particles from the solar wind or radiation bombard rocky surfaces, sputtering off the sodium.
Although Enceladus is mostly rock, the moon’s surface is icy, preventing much sodium from escaping via solar wind or radiation, Lunine says. Instead, the only way sodium can exit Enceladus is for the element to escape from the moon’s interior. And for that to happen, liquid water is required, he asserts.
“A liquid water layer or pocket in contact with the rock, which is deep below Enceladus’ surface, will acquire sodium from the rock — essentially leaching the rock,” he says. If the source of Enceladus’ south polar geysers, discovered by Cassini in 2005, is indeed liquid water, then the geysers will transport the dissolved sodium into space. As the geysers reach Enceladus’ frigid surface, the water freezes and some of the sodium remains trapped within the newly formed ice crystals. Like a frozen ocean spray, the geysers spread the salty ice particles into Saturn’s E ring.
Cassini researcher Roger Yelle of the University of Arizona has a different view. “The surface of Enceladus is not pure water ice. We just don’t have a good idea of what the other components are,” he notes. “There could be a small amount of sodium in the minerals in the surface layer.” The sodium detected in the E ring could have come from that material on the moon’s surface, rather than from its interior. “I don’t believe that you can say that the detection of sodium [in the E ring] implies that it came from a sub-surface ocean” on Enceladus, Yelle says. “So, let’s not run around crazy-like claiming the likely detection of life because sodium was found in dust particles in the Saturn system.”
Another complicating factor is that studies from Earth, using large telescopes such as the Keck Observatory atop Hawaii’s Mauna Kea, have not found any sign of sodium in the E ring. Nick Schneider of the University of Colorado at Boulder reported the lack of sodium last December in San Francisco during a meeting of the American Geophysical Union.
But such studies, notes Kempf, can only detect sodium in its gaseous form, not the solid sodium in the frozen ice particles. He maintains that the bulk of the sodium in the E ring lies in the solid phase recorded by Cassini’s cosmic dust analyzer.
But depending on which of two competing processes dominates in the E ring, Schneider’s nondetection might still pose a problem, says theorist Andy Ingersoll of the California Institute of Technology in Pasadena. Over time, he notes, the ice particles in the E ring will exit, pushed out by the pressure exerted by radiation or by collisions between particles. If this happens quickly enough, the solid sodium would never vaporize but would stay embedded in the ice even as it exits. It would be replenished by new material from Enceladus’ plumes.
However, it’s possible that charged particles bombarding ice crystals in the E ring could liberate some of the solid sodium, turning it into vapor, before the crystals have a chance to exit. In that case, Schneider’s team ought to have detected gaseous sodium. “It’s controversial which of these processes wins out,” says Ingersoll.
For now, says Lunine, “one just has to acknowledge that the two observations potentially might be in conflict.” The missing link, he adds, “is whether the sodium in the E-ring particles really came from Enceladus.”
It would be a stronger argument, Lunine says, had Cassini’s ion and neutral mass spectrometer found sodium when it flew through the plumes this past March. The instrument, which looked for sodium only during short intervals of the flyby, did identify an array of organic compounds in the plumes that could support life. The spectrometer will look again for sodium when it flies through the plumes on October 9, says Hunter Waite of the Southwest Research Institute in San Antonio.
Kempf’s refined estimate is that sodium salts account for between 0.5 percent and 2 percent of the mass of the E ring grains, the team reports in the Nature paper.
Assuming both Kempf’s and Schneider’s observations are correct, any reservoir of water inside Enceladus would have a low salt contact and woud not be in sufficient contact with the moon’s rocky core to leach much salt, Lunine says. “It has to leach some, given the detection” by Kempf and collaborators, he adds. “I could imagine a marginal case where pockets of liquid water today don’t have strong contact with the rock core, even if they did in the past.”