Seth Shipman recorded a movie in DNA — and that’s just the beginning
He is developing tools that may reveal hidden biological processes

Seth Shipman builds new biological tools that could be used to answer big questions.
Lauren Bayless/Gladstone Institutes
Seth Shipman is a magpie of biological innovation. He collects useful parts — from bacteria, nerve cells, reams of genetic data — and transforms them into tools that do amazing things.
One of his best creations so far is a collection of living bacterial cells with DNA that carries an iconic movie of a running horse. Recording images, or any other information, in the genetic material of living cells isn’t just for entertainment; it will give scientists views of processes that are usually hidden.
Imagine designing record-keeping cells capable of eavesdropping on the cellular destruction that precedes dementia in the brain. Or monitoring the elaborate genetic instructions that tell a brain cell how to develop. Or even seeing the exact moment when cellular missteps begin to create a disorder such as schizophrenia.
Scientists can’t do any of this yet. But Shipman, 36, is patient. “If you’re worried about what you can do right now, it’s hard to take a big step forward,” says Shipman, a biotechnologist at the University of California, San Francisco and the Gladstone Institutes, a nonprofit research organization on the UC San Francisco campus. To move forward often requires a pause, a careful reckoning to examine your tools and look around a bit, Shipman says.
His willingness to change perspective and cross disciplines — neuroscience, microbiology, engineering and even art — is unusual among scientists, says Roger Nicoll, a neuroscientist who oversaw Shipman’s Ph.D. work at UC San Francisco. “I get really antsy when I get outside of my comfort zone,” Nicoll says. “He has no comfort zone.”
Shipman combines “this ability to step back and get deep insight with an incredibly high level of rigor to pursue that vein of gold that he comes upon,” Nicoll adds. The bacterial movie feat, for instance, came from Shipman’s frustration with a lack of good tools. He wanted to monitor genes’ behavior inside cells as time passes, but one of the only ways to track that behavior requires killing the cells. “That destruction is something that’s really incompatible with something that happens over time,” he says. So instead of hammering away with the wrong tools, he backed up and thought about what the ideal tool would look like.
The perfect system would unobtrusively monitor cellular events from the inside and provide a record of those activities. During a postdoc at Harvard University, Shipman and colleagues figured out how to best use the gene-editing tool CRISPR to get bacterial DNA to accept foreign snippets, a technique described in 2016 in Science. Once that was achieved, “it was off to the races,” he says.

After discussions with his Harvard benchmate, artist and biologist Joe Davis, Shipman decided to transfer the instructions for the images of a running horse as a nod to the images’ creator, early technologist Eadweard Muybridge. In the late 1800s, in part to settle a debate about whether running horses are ever fully airborne, with no feet touching the ground, Muybridge created a series of horse images that captured the motion. The choice “just seemed to fit,” Davis says.
The response to the running horse movie was big. “I’ve never had a reaction to a paper like that,” Shipman says. Scientists were enthralled with the prospects of the technology. Journalists were enchanted with the idea of a movie embedded in DNA. Even Muybridge historians got excited.
The ability to put external information into living cells, in the right order, described in 2017 in Nature, brought Shipman a step closer to his ultimate goal: to build a tool to record sophisticated cellular information inside the DNA of living cells and reveal biological processes that have remained mysterious. Now Shipman’s team is working on another big part of the problem: coaxing the cells into recording information on their own.
“Seth has pioneered some of the very early and important work in this area,” says synthetic biologist Harris Wang of Columbia University, whose lab also works on cellular recording methods. “He’s going to push the advancement of this field.”
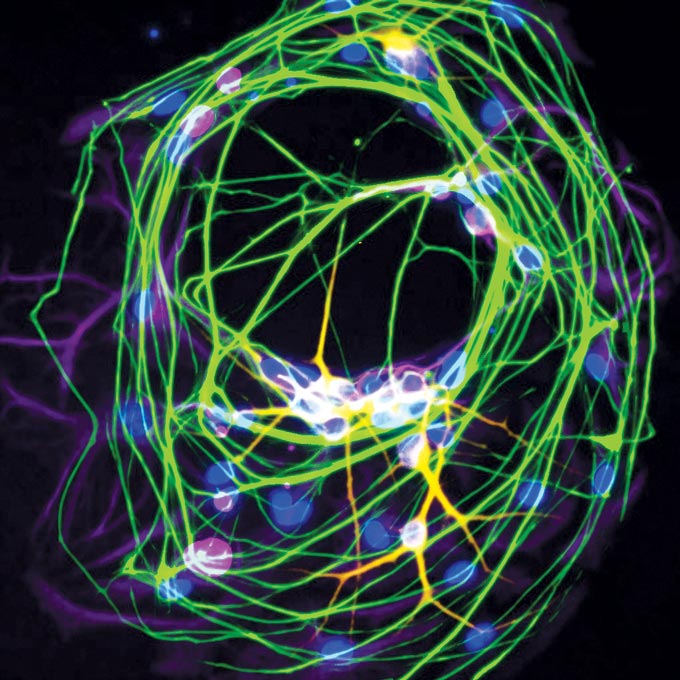
When Shipman and Davis worked side by side at Harvard, they had wide-ranging, boundary-pushing talks. “I could float really crazy ideas,” Davis says, “and he could float really crazy ideas.” Davis mentions the “zen neuron,” a single nerve cell that they grew alone in a dish. With no nearby neurons to send tendrils toward, this unusual cell connected to itself.
Now in his own laboratory at UC San Francisco and Gladstone, Shipman is still cultivating neurons. Many scientists try to figure out how cells form their complex connections by breaking these connections. “Instead of taking something that works and breaking it, we make something, and try to impose order on it,” Shipman says.
His pared-down system, made of one to five neurons growing on a single “island,” offers a way to test the rules governing the shape of more elaborate networks in the brain. Ultimately, the ability to steer cells to form the right connections might become the basis of stem cell therapies for brain disorders. Perfectly programmed neurons could seamlessly replace the cells lost in neurodegenerative diseases.
Where Shipman’s tinkering might lead next is anybody’s guess. “There is no doubt that he will do very well, but you have no idea where it’s going to go,” Nicoll says, “which is great.”








