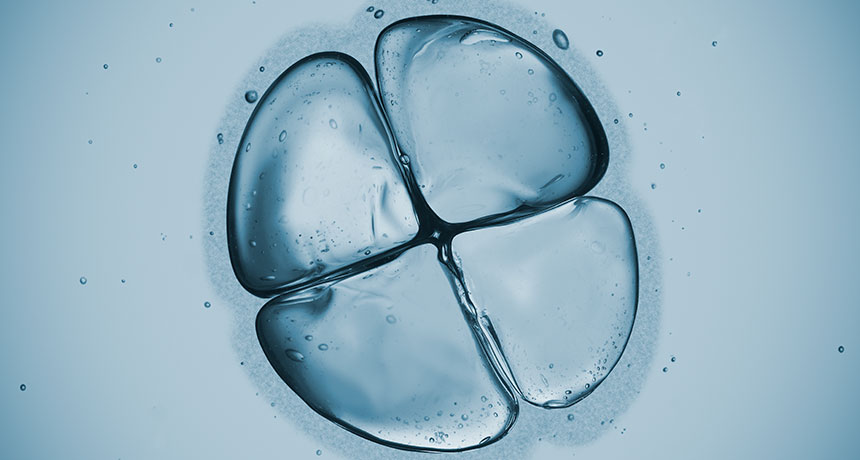
IN THE BEGINNING Early embryos (a four-cell embryo shown) from mice and humans look the same on the outside, but gene activity studies show some big differences under the hood.
Ugurhan Betin/iStockphoto

IN THE BEGINNING Early embryos (a four-cell embryo shown) from mice and humans look the same on the outside, but gene activity studies show some big differences under the hood.
Ugurhan Betin/iStockphoto