Weapon of bone destruction identified
Enzyme finding may aid hunt for new anticancer therapies
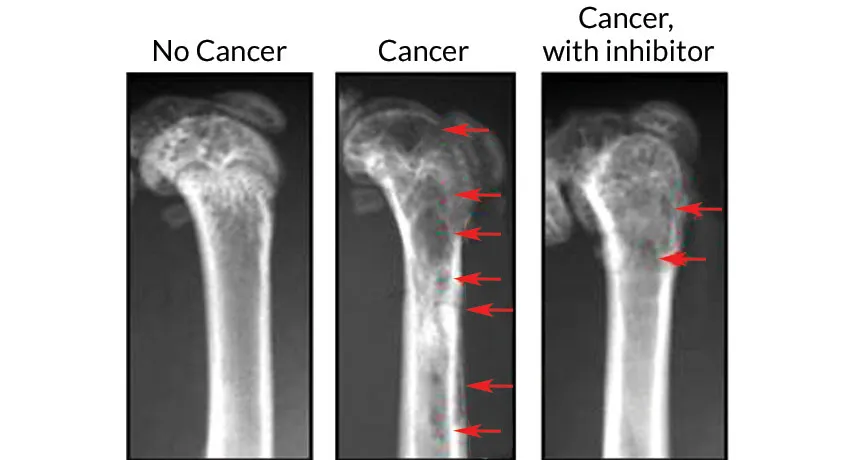
NO HOLES BARRED Mouse femurs (shown in X-rays) with multiple myeloma growing in the bone marrow develop holes and weak spots (middle, red arrows). Treating mice with a drug that inhibits an enzyme responsible for kick-starting the destruction prevents much of the bone loss (right).
H. Liu et al/Science Translational Medicine 2016