Forget soft, cushy digs. Bacteria like to set up house and home on stiff surfaces.
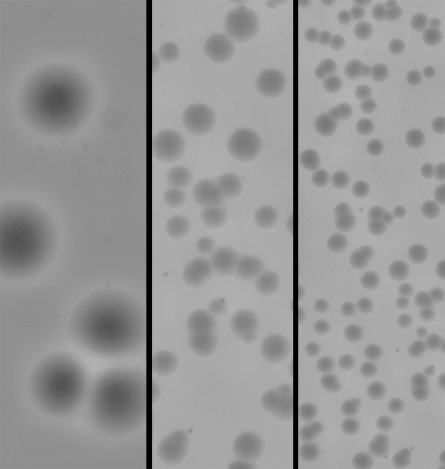
Bacteria form more colonies on rigid surfaces, a team of researchers report in the May 2 Biomacromolecules. The trait could be exploited to improve designs of bacteria-resistant medical implants.
When free-floating bacteria come into contact with surfaces, they sometimes attach and form a biofilm — a layered mass of bacteria that ooze polymers that create a protective matrix. The sticky structure helps glue the bacteria in place. Surrounded by the matrix, the bacterial biofilm can go on to cause antibiotic-resistant infections.
Biofilms often colonize medical devices like joint replacements and catheters. Infections from biofilms cost billions of dollars each year, says E. Peter Greenberg, a microbiologist at the University of Washington in Seattle who was not involved in the study. “Once that infection has started, you can’t cure the infection any other way than to remove the device,” Greenberg says.
Before bacteria can build a biofilm, they first have to gain a foothold on the material, says Krystyn Van Vliet, a materials scientist at MIT. So Van Vliet and her colleagues tried to pinpoint factors that help the bugs stick in the first place. The team added two troublesome bacteria, Escherichia coli or Staphylococcus epidermidis, to test surfaces, and watched bacterial colonies grow. Stiffer materials grew more colonies for a given amount of surface area, with a material 100 times stiffer producing colonies 100 times more dense, says Michael Rubner, a materials scientist at MIT who was also involved in the study.
The team created multiple surfaces that were chemically identical but varied in stiffness. To fine-tune the stiffness, the team coated glass slides with many layers of positive and negatively charged polymer molecules. “You dip a glass slide into the positive polymer, then rinse it a few times in water, then dip it again in the negative polymer,” says Van Vliet. The positive and negative solutions then bind to each other, she says.
The more acidic the solution, the less tightly the positive and negative polymer layers cling to each other. So when dipped in water, the more acidic polymer solutions grew squishier by swelling like a sponge. Polymers closer to neutral stuck together more, didn’t swell as much when water was added, and were firmer as a result.
Just making implants of soft material won’t be enough to fight off infection. “We know from past studies, that if you’re thinking of making an antibacterial coating, this is not going to be sufficient,” Rubner says.
Greenberg agrees. “They don’t have a material now that you would want to replace all implanted devices with because it resists colonization,” He says. And molecules from the body quickly coat implant surfaces, and bacteria usually glom onto those molecules, rather than the surface itself. So how well the material could resist a bacterial onslaught in a clinical setting is an open question, he says.
To be useful, implants might need to combine existing anti-microbial treatments with squishier surfaces, Rubner says. “We want to look at both and see if it provides an extra one-two punch for keeping bacteria off surfaces.”
Right now, no one knows what makes bacteria stick better to rigid surfaces, but the team has some ideas. The bacteria in the study sport long rods called pili, which are capped by sticky molecules. Other research showed that these molecules bind more tenaciously to surfaces when the pili are tugged on, Van Vliet says. “It’s a little bit like Velcro,” she says. It’s possible the same mechanism helps bacteria settle down and colonize rigid surfaces better.






