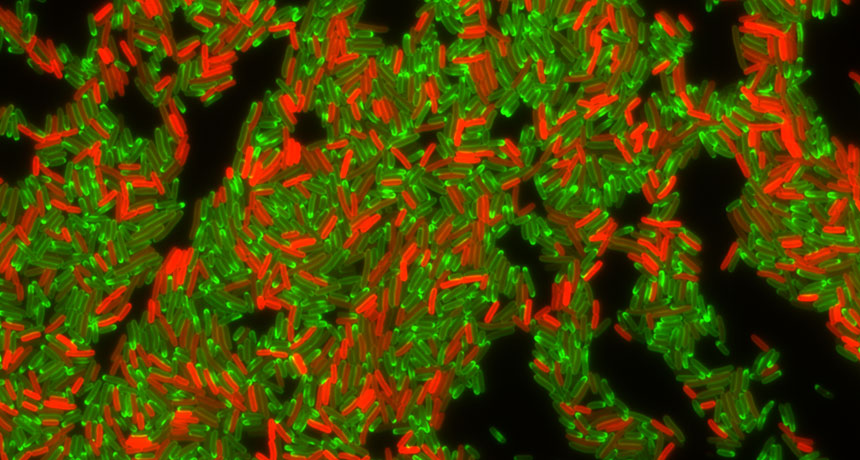
PUMPED OUT E. coli bacteria can become resistant to the antibiotic tetracycline by making a protein that pumps the drug out of a bacterial cell. Here, resistant bacteria that make the pump protein glow red, while bacteria that don’t make the pump (green) are filled with the antibiotic.
Sophie Nolivos






