Cartilage Creation
New joint tissue could keep people moving, reducing need for knee or hip replacements
Cartilage, the shock absorber of the body, has been bearing the brunt of a modern lifestyle.
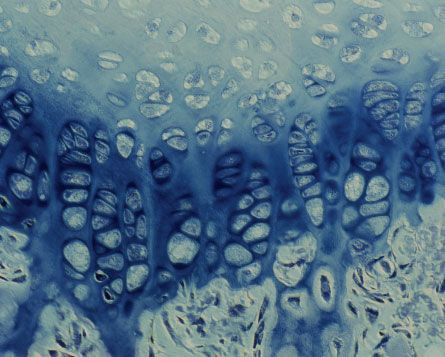


This nerveless connective tissue allows bone to glide over bone without any repercussions — most of the time. But human cartilage evolved in an earlier age, in ancestors who lived shorter lives, carried less body weight and roamed an unpaved world. Nowadays, cartilage takes a constant and prolonged beating from which it has poor capacity to bounce back.
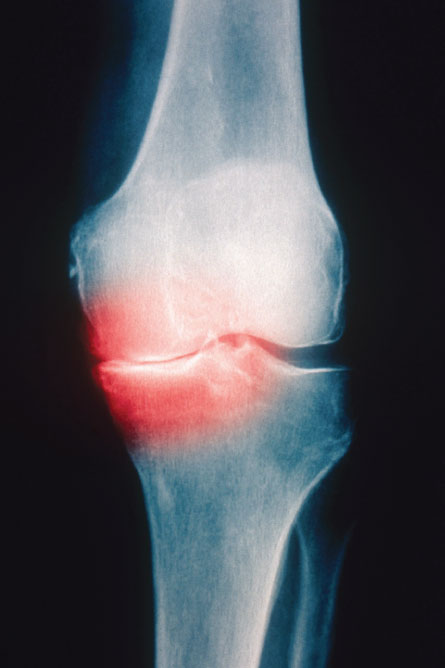
It’s not a good scenario for an aging population. As the go-between tissue in joints, cartilage can handle only so many jolts and jars before something has to give. When the daily grind wears the tissue down, or it gets damaged by more abrupt injury, the bones’ nerve cells become exposed. Movement can lead to a painful zing, the hallmark of osteoarthritis, which now affects more than 27 million people in the United States. In addition to pain, osteoarthritis shows up as stiff joints, cracking sounds, inflammation and bone spurs.
What’s worse, when it comes to cartilage damage, there is no safety net. Cartilage manufacture depends entirely on one type of cell, chondrocytes, and these tiny cartilage factories multiply less and less with advancing age. They fall behind in making new cartilage to repair defects and sometimes respond to injury or long-term pounding by giving up and dying off.
No wonder some orthopedists consider cartilage regeneration the holy grail of their field. Confronted with hordes of gimpy people living longer lives, orthopedists have used surgery to clean out damaged joints, braces to stabilize a wobbly gait and artificial knees and hips to replace damaged bone ends, a last resort against osteoarthritis. Everything short of new cartilage.
But now, with the help of stem cells, a new generation of bioengineers are coming close to cracking the code for cartilage regrowth. Stem cells have yet to choose a career path, a characteristic that makes them attractive future cartilage-creators. The blue sky version of the stem cell approach goes like this: Stem cells are extracted from a patient, geared up to become chondrocytes, wrapped in a favorable mix of compounds and then inserted into damaged joints. The cells take it from there. Voilà: neo-cartilage.
Scientists have recently pinpointed prominent proteins needed to keep stem cells on track to becoming cartilage-making chondrocytes and have even devised nanosized polymer scaffolds on which these stem cells can start growing cartilage. The cells seem to behave better if surrounded by molecules found naturally in healthy cartilage, and some research suggests scaffolding derived from cartilage itself might deliver much-needed biochemical prompts.
While still in the experimental phase, the stem cell strategy is gaining ground: More than a dozen clinical trials using stem cells as cartilage regenerators are under way or planned in Norway, Spain, Iran, Malaysia, France and elsewhere.
Tissue engineering and cartilage regeneration were at “point zero” in the 1970s, says Wan-Ju Li, a tissue engineer at the University of Wisconsin–Madison. “Now the technology is more mature. All together, the field is getting very rich and very interesting.”
Getting glassy
Researchers aren’t interested in making just any cartilage. They want the kind that caps the ends of long bones, such as those in the legs. This type, called hyaline cartilage, is distinct from the bendable kind in the ears or the fibrocartilage found between vertebrae. Hyaline cartilage is slippery, glassy, elastic and smooth. Picture the tough gristle at the rounded end of a ham bone. It is everything humans would want to cap a bone in a weight-bearing joint, allowing them to move about like pain-free machines.
But past attempts to repair hyaline cartilage through regeneration have come up short. For decades, the surgical approach for a damaged joint has been to clean out frayed cartilage and, sometimes, drill tiny holes into the worn ends of the bone. The holes allow blood and stem cells from bone marrow to leak out and patch the injury.
In theory that should work, but the stem cells seeping through the holes lack focus. “Those stem cells that come out are confused,” says John Sandy, a biochemist at Rush University Medical Center in Chicago. “They’re not getting the right signals.… So they hit the middle road.” They make fibrocartilage, a poor substitute for hyaline. In a recent study, only two-thirds of athletes receiving this “microfracture surgery” following injury showed good results, and only half maintained their original level of play for several years.
Doctors have also transplanted living chondrocytes onto worn-out bone ends. Genzyme, a biotech company based in Cambridge, Mass., offers an off-the-shelf kit for this procedure. It requires taking thousands of live chondrocytes from healthy cartilage elsewhere in the body, culturing the cells in a dish to expand their numbers and packaging them with other products for insertion into the trouble spot. Called autologous chondrocyte implantation, the procedure has outperformed microfracture surgery in some studies, but some patients need follow-up surgery and a nine-year study of implantation patients found that 30 percent didn’t improve. The trouble may arise because mature chondrocytes lose their ability to produce cartilage if expanded through too many generations, Li says.
Stem cells have an advantage there. Like newly hired employees, they should have plenty of productive years in store. Key for scientists is finding a reliable way to teach these blank slate cells to become hyaline-producing chondrocytes.
Cellular prompting
Scientists can round up starter stem cells from all over the adult body. Those found in connective tissues such as cartilage, tendons and the synovial membrane that forms a sac enveloping joints have the potential to make good chondrocytes. But so might some less-obvious choices, such as stem cells from bone marrow, fat tissue and discarded umbilical cords.
Before sending them into the fray, though, scientists need to give the cells every possible advantage, nudging them toward chondrocytic behavior. In the lab, the cells are typically mixed with a brew of natural chondrocyte-promoting compounds and then seeded onto a scaffold that encourages growth and cartilage production. The seeded scaffolding is later inserted into a cartilage defect in a patient.
An optimal recipe for the cell-directing brew is still a mystery, but scientists have identified several possible ingredients.
Essential to the mix are compounds called growth factors. The best-studied is TGF-beta, which is good at jump-starting a stem cell to act like a chondrocyte and produce cartilage, Sandy says. But TGF-beta can’t work alone; relying too heavily on TGF-beta, for example, can steer a stem cell toward making fibrous tissue, rather than the resilient hyaline cartilage, he says.
Recent research has focused on another growth factor called FGF-2. Li and Wisconsin colleague Andrew Handorf reported last year in PLoS ONE that treating stem cells with FGF-2 primed them to become hyaline-making chondrocytes.
FGF-2 activates a compound called Sox9 in the stem cell, which in turn switches on the production of two main components of cartilage, type 2 collagen and aggrecan, Li says. FGF-2 might be best used before cell differentiation, the point at which a stem cell becomes a mature cell with a specific role, he says. Then other growth factors, including TGF-beta, could push the cartilage-making process along. A 2010 review lists a dozen growth factors that affect stem cells’ ability to differentiate into chondrocytes.
But these growth factors must be wielded carefully. Ming Pei, an orthopedic surgeon and cell biologist at West Virginia University in Morgantown, says even FGF-2 can cause the resulting chondrocytes to swell in size and swing toward bone making. His team has found that combining FGF-2 with certain booster proteins, as well as cartilage tissue with its own chondrocytes removed, helps increase the numbers of stem cells in the lab and, later, keeps the cells on task.
Another booster compound called kartogenin can steer stem cells directly toward making components of hyaline cartilage, researchers report in the May 11 Science (SN: 5/5/12, p. 10). Kartogenin inhibits a protein called filamin A in the stem cells, an action that unleashes other compounds that switch on genes that ultimately trigger cartilage creation.
This molecule shows promise, say Joan Marini of the National Institutes of Health in Bethesda, Md., and Antonella Forlino of the University of Pavia in Italy. “Stimulating the differentiation of one’s own stem cells by means of an easily deliverable chemical compound would be more advantageous than using conventional drilling and microfracture techniques,” they write in the June 28 New England Journal of Medicine.
A protein called vimentin takes a different route to reach the same objective. To differentiate into a chondrocyte, a stem cell must take on a round shape, says Rocky Tuan, a tissue engineer at the University of Pittsburgh. He and his colleagues found that vimentin nudges bone marrow stem cells toward becoming rounded like chondrocytes. Extra vimentin also boosts genes instrumental in making type 2 collagen, Tuan’s team reported in 2010 in the Journal of Cellular Biochemistry.
The blend of compounds required to create a good hyaline-making chondrocyte may ultimately hinge on the choice of stem cell itself, Pei says. He proposes that stem cells derived from the synovial membrane have an advantage by already coming from a joint. In fact, synovial stem cells manufacture a substance, a type of matrix, that seems particularly valuable.
Pei’s team mixed the matrix made by synovial stem cells with FGF-2 in a low-oxygen environment. That combination, when added to other synovial stem cells, enabled those cells to ramp up their numbers. It also provided them with a favorable “niche,” a microenvironment amenable to chondrocyte formation, the researchers reported last year in Tissue Engineering, Part A.
Optimizing these conditions improves the niche and helps the stem cells thrive, in part because they are free of stress. Pei’s approach limits reactive oxygen species, unstable molecules that damage tissues, keeping the stem cells comfortable. Growing outside of niches, Pei says, “stem cells bear the stresses of the environment and lose their proliferation capacity. They become old.”
A place to reside
Efforts to expand stem cell numbers and steer them to become chondrocytes go for naught if the cells can’t hold together long enough to form a cartilage patch. A stable home would be a replica of what scientists call chondrocytes’ extracellular matrix, the elastic web of cartilage naturally surrounding them in the body.
About a decade ago, Li and Tuan became interested in building a synthetic scaffold resembling the naturally occurring one, onto which they could seed stem cells. While still at the National Institutes of Health, they used a process called electrospinning to cast nanofibers of polymer into a structure resembling cartilaginous matrix. Those first webs have since been improved and made into a biodegradable cartilage scaffold.
“It’s easy to fabricate and there’s no batch-to-batch difference,” Pei says.
Li notes that the polymer, built to degrade after six to 12 months in the body, has strong mechanical properties that keep stem cells together, giving the cells time to weave their own matrix of cartilage.
In 2009, Tuan and Li tested their scaffold, seeding it with human stem cells to create a patch that was then inserted into pigs with cartilage damage in their knees. Some pigs received scaffolding seeded with mature chondrocytes. The researchers allowed the pigs, with some restrictions, to put weight on the knees almost immediately, since routine compression is the norm for cartilage.
After six months, the stem cells had grown into chondrocytes that made hyaline cartilage that outperformed the fibrocartilage made by the mature chondrocytes.
“It was glassy cartilage,” Li says, “with good mechanical properties.”
In an alternative approach, other researchers are testing the scaffold potential of a cartilage matrix obtained from cadavers. The scientists remove the cells and use the rest. Though that work is in its early stages, Pei says the natural matrix offers some useful biological cues for stem cells that a polymer doesn’t.
Still other teams are instead using a natural adhesive called fibrin glue as scaffolding material. Doctors at Cairo University combined bone marrow stem cells with fibrin glue and blood platelets — a healing aid that produces the growth factor TGF-beta — and placed the mix in the cartilage-damaged knees of five patients. A year later, all reported improvement.
Regardless of exactly which blend of boosters and scaffolding work best, Li is optimistic that a stem-cell strategy, once fully researched, will succeed in making cartilage where aging chondrocytes have failed. “We believe we can do better,” he says.
Early adapters
Once that goal is achieved, one of the big challenges will be to determine which patients would benefit most from the treatment. Much osteoarthritis seems to unfold over a lifetime, but many patients develop it from hyaline cartilage damage traceable to an injury. If cartilage regeneration reaches the clinic, treating these patients early could prevent further loss of cartilage and prevent osteoarthritis from overtaking the joint, says David Felson, a rheumatologist at Boston University School of Medicine.
Cartilage is made to handle stress and compression, but every tissue has its limits, and knee injury increases by sixfold the likelihood that a person will develop osteoarthritis. Felson says his team has found evidence that injuries to the knee structures “probably account for a great majority of osteoarthritis.”
But early detection isn’t easy since many people ignore or underestimate their injuries. Years can pass before X-rays and other scans show two bones rubbing together, a sign of painful cartilage loss and budding osteoarthritis.
Carla Scanzello, a rheumatologist at Rush University Medical Center, and colleagues reported last year that inflammatory molecules that gradually destroy cartilage flood an injured joint and leave a telltale signature long before symptoms of osteoarthritis arise. Understanding these markers of inflammation “might help us target patients,” she says.
For the moment, that targeting would involve anti-inflammatory drugs to limit the cartilage degradation. But in the future, a stem cell treatment might dramatically reduce the number of people who end up needing a joint replacement, says Dobrila Nesic, a molecular biologist at the University of Bern in Switzerland.
An artificial knee or hip can last 10 to 15 years, Nesic says, and a person can get two (per joint) in a lifetime. “If you’re 60, no problem,” she says. “If you’re 40, your lifetime before wheelchair is 20 to 30 years.”
Researchers agree more work is needed to bring stem cell–based cartilage regeneration to the clinic. Li suspects the technical problems might be solved in the next five years or so, with another five years needed to sort out regulatory and insurance issues.
He has students who are surgeons, still busy learning how to replace knees and hips. “I was joking with them,” Li says, “saying, ‘You guys are going to have to find a new job soon.’”







