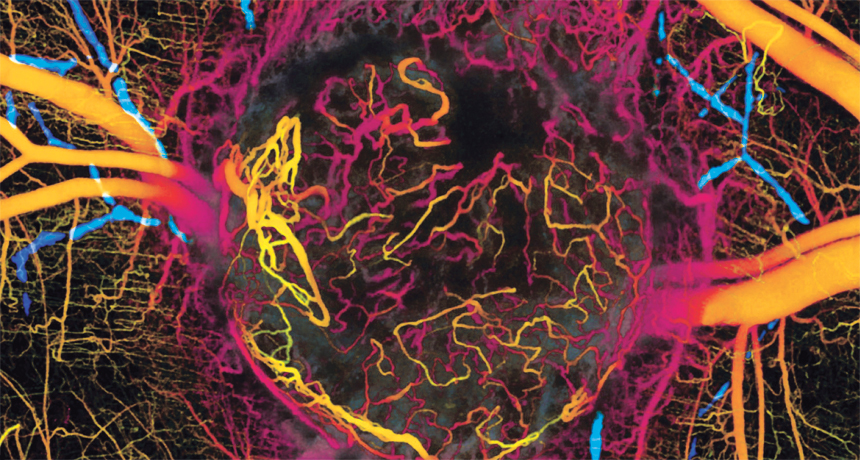
UNHAPPY HOME A breast tumor grown in a mouse is lined with twisted and leaky blood vessels (orange and pink). Lymph vessels (blue) surround the tumor. New studies show the importance of a tumor’s environment in determining how aggressive a cancer will become.
B.J. Vacok et al/Nature Medicine 2009
It’s what’s inside that counts, right? Not so when it comes to cancer, says Mina Bissell.
True, gene mutations inside a cell help determine whether it will become a tumor. But, according to Bissell, a cancer researcher at Lawrence Berkeley National Laboratory in California, the neighborhood immediately surrounding the cell is just as important.
Cells in healthy tissues nestle tightly against each other like row houses. These rows sit on top of, and are surrounded by, a dense scaffolding of proteins known as the extracellular matrix. Blood vessels act like water pipes to bring in nutrients; lymph vessels drain away waste like sewer lines. In thriving neighborhoods, any trash that builds up within a cell doesn’t spill out to pollute the rest of the area. If it does, the neighbors clamp down like a strict homeowner’s association to contain the problem and bring the cells on the block back into line.
In crumbling localities, however, cells lose touch with their neighbors or run amok after falling in with the wrong crowd.
Bissell’s experiments over four decades have repeatedly demonstrated that a cell’s neighborhood — its microenvironment — is a key influence on whether cancer will develop and how bad it will get. In the past 5 to 10 years, many other cancer researchers have also found evidence to support that view.
Their research is transforming thinking about cancer and what to do about it. Bissell and others are convinced that by properly managing a tumor cell’s microenvironment, they can make cancer something people live with, not die from.
“We really should be able to keep cancer as dormant disease,” Bissell says.
Her ideas are the antithesis of what most people have come to believe about cancer: Genes determine how nasty a tumor will be and the only way to deal with the disease is to eradicate it. Many cancer researchers have focused on the mutations that turn upstanding cells into cancerous thugs. Some even view those genetic changes as the sole determinant of cancer.
The view of genetics as cancer fate made headlines earlier this year when Angelina Jolie announced that she had her breasts removed because she carries a mutation in the breast cancer associated gene 1, or BRCA1. The odds are clearly against women with Jolie’s family history (her mother died of breast cancer) and genetic makeup. On average, about 8 of every 100 women will develop breast cancer by age 70. But 50 to 70 of every 100 women with BRCA1 mutations will get the disease before age 70, and usually at much younger ages.
Bissell sees a different story in those numbers. She points to all the women with a BRCA1 mutation who never develop cancer. Plus, she adds, the gene is instrumental for helping repair damaged DNA in every cell of the body. Yet most carriers of the faulty gene develop either breast or ovarian tumors. “So why don’t you get pancreatic cancer or brain cancer?” Bissell asks. For that matter, “why doesn’t the entire breast or the entire body turn into a tumor?”
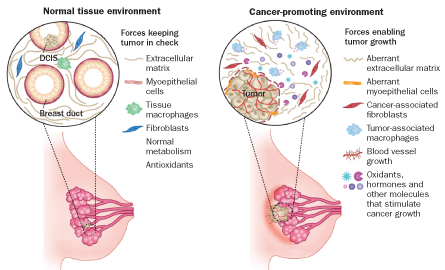
The answer, she and a growing cadre of researchers contend, is that mutations aren’t alone in determining whether breast or other cancers will arise, spread and kill. Controlling cancer is a community effort, with nearly every denizen in a cancer cell’s neighborhood playing a part.
Missing protection
Breast cancer often starts when mutations hit the epithelial cells that line the milk ducts in the breast. Varieties of epithelial cells are found in other organs, too, such as the lining of the intestines, kidney, stomach and pancreas. They are in ducts and glands, line the air sacs of the lungs and form skin. They are also the type of cell from which most cancers arise. In the breast, those mutations initially cause a noninvasive precancerous state known as ductal carcinoma in situ, or DCIS. About 20 to 30 percent of women with DCIS will develop invasive breast cancer.
Kornelia Polyak of the Dana-Farber Cancer Institute in Boston and her colleagues thought that the epithelial cells that produce DCIS must be fundamentally different from the ones that would break out of the duct and become dangerous invasive breast cancers. But close examination showed that DCIS cells and invasive cancer cells carry the same sorts of mutations and grow similarly in laboratory dishes, she says. What changes between not-yet-cancer and cancer is the behavior of neighboring cells, Polyak and others have found.
She has evidence that myoepithelial cells, which form a layer next to the epithelial cells, are important for keeping DCIS inside the ducts. If those myoepithelial cells fall down on the job, the cancer cells can break out and form dangerous tumors.
About half the cells that make up most solid tumors are a mix of noncancerous cells known collectively as stroma. (In pancreatic cancer, stromal cells can make up about 90 percent of the tumor.) The stroma of a breast tumor includes blood vessels, a wide variety of immune cells, fat cells, cells called fibroblasts and the mesh of proteins called the extracellular matrix that give tissues their shape.
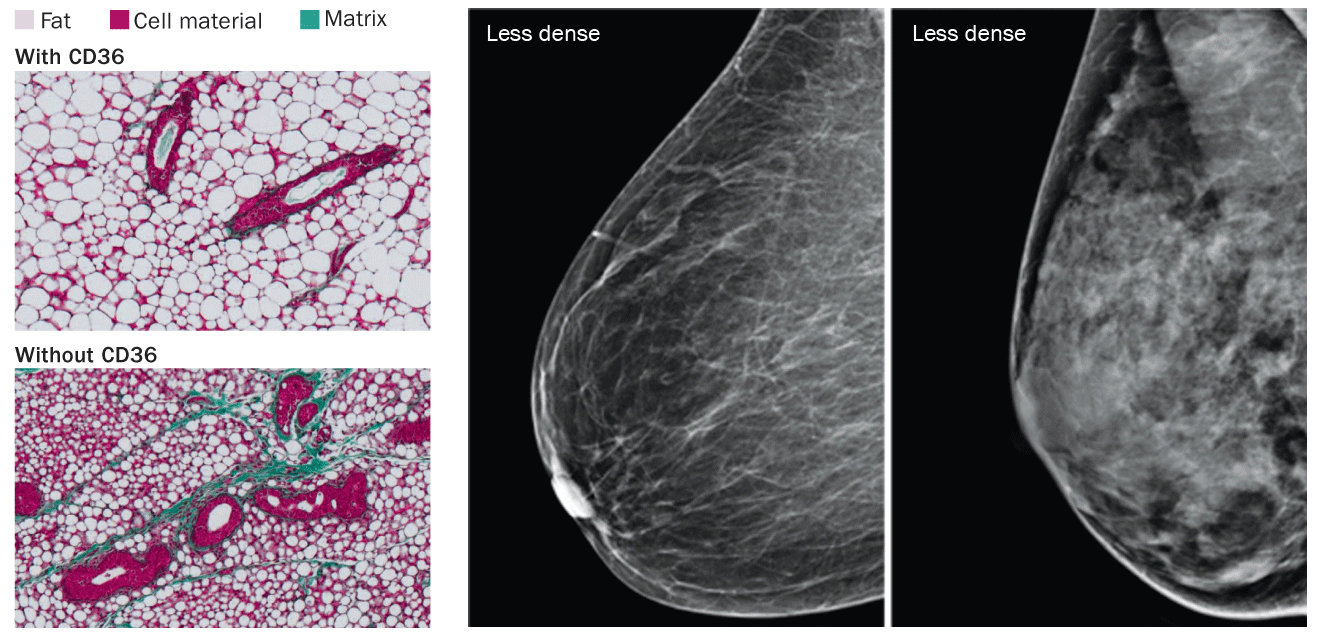
Delicate strands of extracellular matrix proteins such as collagen and elastin are barely visible on mammograms of healthy breasts, says Thea Tlsty, a molecular pathologist at the University of California, San Francisco.
But in some women’s breasts, collagen, elastin and other extracellular matrix proteins form thick ropes of connective tissue that appear as stringy white material on a mammogram. Those women are said to have dense breasts and are at increased risk of developing breast cancer.
Tlsty and her colleagues discovered that a protein that helps strike a balance between making fat and producing connective tissue is partly responsible. The protein, called CD36, exists on many types of cells. CD36 is a busy molecule, regularly making contact with a wide variety of proteins in the extracellular matrix and on neighboring cells. Among its many roles, CD36 controls processes that can promote healthy breast tissue. When missing, it leaves behind an environment that allows cancer to grow, Tlsty and her colleagues reported last year in Cancer Discovery.
Rosa Anna DeFilippis from Tlsty’s team studied breast tissue fibroblasts from women who had undergone breast reduction surgery or who had had tumors removed. Fibroblasts are cells that help construct connective tissue, and are some of the main builders of the extracellular matrix components that contribute to breast density.
Women with low breast density (more fat than connective tissue) had more CD36 on their fibroblasts than women with high breast density. Fibroblasts with low levels of CD36 pump out more of the stuff, like collagen, that creates the whiteness on mammograms, Tlsty says. Lower levels of the protein also spark other cancer-stimulating changes. Immune cells that don’t make CD36 transform from tumor fighters into cancer promoters. And wiping out CD36 from blood vessel cells removes the brakes that normally keep vessel growth in check, the researchers found.
Tlsty’s group has seen these procancer changes in the stroma occur before there’s a tumor. “That’s amazing,” she says. It suggests that the stroma goads tumor growth. Everybody else thought that the tumor itself caused the abnormal growth of connective tissue, she says. Perhaps correcting the CD36 levels could eventually be both therapy and prevention for breast cancer, she says.
No place like home
This sort of data comes as no surprise to Bissell.
After all, her lab demonstrated that placing breast cells in a laboratory setting that reminds them of home is enough to get the cells to remember how to do one of their primary jobs: producing the proteins that go into milk. And altering the way breast cancer cells interact with some proteins in the extracellular matrix coaxes cancer cells to behave like normal cells, her team showed in 1997.
Since then, her lab has piled up dozens of papers that detail how cancer cells interact with different aspects of the microenvironment. The complexity she revealed is part of the reason it took some scientists time to appreciate Bissell’s argument, she says. “What I was saying was not so easy to demonstrate in one paper.”
What’s helped her move the work forward is growing human cells in three-dimensional gels that mimic growth in a real tissue. Many researchers grow mouse cells in two-dimensional dishes that produce less realistic conditions.
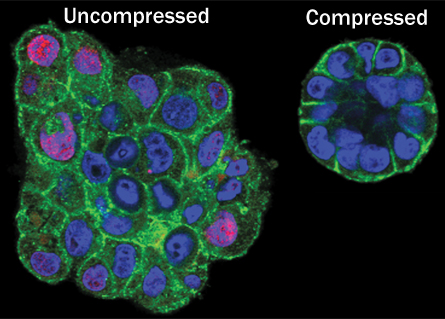
The 3-D system has revealed that mechanical forces exerted by cells pressing up against each other can have a calming effect on cancer. Bissell and collaborators at the University of California, Berkeley demonstrated last year that putting breast cancer cells under gentle pressure, akin to what they might encounter growing in a healthy breast, causes them to reconnect with their neighbors and behave like noncancerous cells (SN: 1/26/13, p. 8). That does not mean that squeezing breasts will prevent breast cancer. It just shows that cells get many different types of cues from their environment about how to behave.
Once a cancer cell has migrated to a new location in the body, it can nestle against blood vessels and lay dormant for years, Bissell and colleagues have found. If the blood vessels remain as they are, the cancer will continue to snooze. But when new blood vessels sprout, dormant cancer cells wake up and new tumors grow, Bissell’s group reports in the July Nature Cell Biology.
“I argue that cancer is not a problem with growth, as the entire field believes,” Bissell says. Instead, it’s a matter of context. Like small-town kids who get lost in the glitz and glam of the big city, cancer cells can find themselves disoriented when their surroundings change. Soon, they are running amok, behaving in ways they never would at home, where their neighbors help keep them grounded. Incubating cancer cells with components of the extracellular matrix puts the cells in a familiar setting so they can remember their identities and job responsibilities, Bissell says. “This is so obvious. Why don’t people get it?”
The message is sinking in, says Suresh Mohla, who heads the Tumor Microenvironment Network at the National Cancer Institute in Bethesda, Md. The institute launched the microenvironment initiative in 2006. By that time, data collected by Bissell and others had become far too compelling to ignore. Now researchers realize that the tumor microenvironment isn’t just one thing, says Mohla. “A tumor is just like an organ,” he says, “It’s a complete functioning unit.”
Emergency responders
Like other cells in the body, a tumor has to find ways to deal with stresses, such as physical pressures, chemical signals and changes in blood sugar and oxygen levels.
The most common response to stress involves making chemicals called reactive oxygen species, which are molecules that bash DNA, proteins, lipids and other components of a cell. Unchecked, these molecules — also known as “oxidants” or “free radicals” — can severely harm or even kill a cell. Most of the time, spillover from stressed cells is mopped up, says Tlsty, but sometimes a small amount can get away and cause damage. Scientists have known for a long time that having too many oxidants raises cancer risks, but the exact mechanism has been murky.
Tlsty and her colleagues are closer to understanding how the process works. Oxidant damage triggers a molecule called Activin A to go to work, the researchers reported last year in Breast Cancer Research. Activin A makes the molecular equivalent of a 911 call, Tlsty says. “It says, ‘we’ve got a problem here, folks. Gather around. Help.’ ”
Among the first to respond to Activin A’s distress call are molecules involved in promoting inflammation, the team found. The responders, such as COX-2 (the molecule inhibited by the anti-inflammatory drug Celebrex), unleash a flurry of messages to rival Twitter spikes following a riot. Those messages trigger an avalanche of changes conducive to cancer.
Shutting down inflammation entirely is not the answer. Inflammation is a necessary part of the body’s ability to heal wounds. When the immune system detects injuries or aberrations such as cancer cells, it sends in immune cells to clean up the mess and stitch broken tissue back together. “That we don’t all walk around with big tumors is proof that the immune system works most of the time,” says Lisa Coussens, a cancer biologist at Oregon Health & Science University in Portland.
The problem comes when the clean-up crew encounters rapidly growing cells that won’t die on command — in short, cancer. The nascent tumor essentially becomes a wound that will not heal. Inflammation becomes a chronic condition and the tumor feeds on chemicals released by immune cells. Cancer also seduces immune cells into acting as escorts, helping it spread throughout the body.
Coussens and her colleagues found that immune cells called macrophages are attracted to tumors under attack from chemotherapy drugs. The macrophages toughen up the tumor, making it less sensitive to the cancer drugs, the researchers reported in the June 2011 Cancer Discovery. Drugs that blocked the macrophages from reinforcing tumors under siege helped restore chemotherapy’s cancer-killing ability.
Researchers are testing other drugs that manipulate the immune-cell component of the microenvironment to see if they can halt cancer in patients. Some medications such as aspirin or other anti-inflammatory drugs could reduce the amount of cancer-stimulating inflammation in tissues. Coussens and colleagues argue in the Jan. 18 Science that combinations of therapies, maybe even including vaccines or fasting, will probably be needed to restore proper immune system balance.
Calming angry tumors
Researchers realized long ago that the blood supply is one aspect of the tumor microenvironment that might be easy to manipulate. Tumors need oxygen and other nutrients to grow, so it makes sense to choke off a tumor’s blood supply and starve it. To that end, drug companies have developed medications that interfere with molecules that stimulate blood vessel growth. Several have been approved for use in various advanced cancers and a few are being tested in patients with breast and other cancers.
Many of the drugs, known as angiogenesis inhibitors, block one of the most potent blood vessel stimulators, a protein called VEGF. But their impact in clinical trials has been mixed. Rakesh Jain, a chemical engineer turned cancer researcher, has been thinking about flow problems for a long time; he did a master’s thesis on water flow in the Delaware River before turning his attention to cancer. “While everyone was working on anti-angiogenesis, I was scratching my head saying, ‘It’s not going to help.’ ” Instead, in 2001, Jain proposed doing the opposite: increasing a tumor’s blood supply and repairing leaky blood vessels with anti-VEGF drugs used in judicious doses.
Open the spigot and feed a tumor — with tumor-starving drugs? “It was not easily embraced,” Jain says of his idea. Many scientists still don’t buy it. But Jain, of Harvard Medical School and Massachusetts General Hospital, isn’t daunted. He lays out the unusual strategy and his rationale in the June 10 Journal of Clinical Oncology.
It’s a matter of balance, he says. His team’s studies show that stromal cells, not just cancer cells, make VEGF. The protein produces vessels that feed the tumor, but the vessels are twisted, leaky and often dead-end unexpectedly. The cancer and stromal cells can crush delicate blood and lymph vessels. The effect is similar to kinking a garden hose or poking holes in it; not much fluid flows through. That makes for an angry, low-oxygen tumor. And lack of oxygen, Jain says, turns on some processes and switches off others to make the tumor resistant to chemotherapy and radiation. It also stimulates abnormal blood vessels.
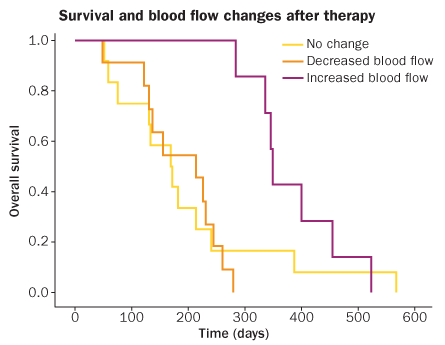
While some studies have shown that anti-VEGF drugs restrict blood and oxygen supplies — think angry tumor — others indicate that anti-VEGF drugs can plug leaks in blood vessels. Jain hypothesized that, at the right levels, anti-VEGF drugs should restore blood flow and allow chemotherapy drugs to penetrate the tumors.
Jain and his colleagues looked carefully at the data from studies in brain cancer patients in which one anti-VEGF drug had no apparent effect on survival. Blood flow stayed the same or decreased in some patients, while others had increased blood flow to their tumors. The people whose blood flow increased lived about six to nine months longer. The drug, produced by a company that Jain has consulted for, has not received approval for widespread use. Jain is hopeful that a cocktail of drugs that open up squished blood vessels and stop leaks could improve cancer therapy in the future.
Seeing the microenvironment as a key player in cancer is finally gaining traction, says Mohla of the National Cancer Institute. And Bissell says she’s close to figuring out what separates a healthy neighborhood from one that would let cancer run wild. She’s already certain of one thing: The answers won’t be neat or simple. “Biology is not clean,” she says. But solving the puzzle of the cancer microenvironment is worth the effort. One day, doctors and patients may be able to transform cancer from a threatening thug to a well-behaved neighbor.







