Steve Haddock remembers every detail about his first ocean encounter with a comb jelly. The open water was a bottomless deep blue. The animal, about the size of a tennis ball, shimmered with bioluminescence. “It was just cruising along like a hover craft,” says Haddock, a marine biologist at Monterey Bay Aquarium Research Institute in Moss Landing, Calif. “Comb jellies are more alien than any aliens people imagine,” he says.



Start with their appearance: The marine animals resemble translucent balloons rigged with flashing, colored lights. Some species glow. When startled, some flash electric blue. Vertical rows, or combs, made of hundreds of iridescent, hairlike cilia run the lengths of their globular bodies (thus the name comb jellies). In some species the cilia are 2 millimeters long — 200 times the length of cilia in other animals — and they beat in coordinated waves, propelling the jellies forward, backward and diagonally in search of prey.
It’s not just their appearance that is wondrous: Slice a comb jelly embryo in two and you get two half-adults that can fertilize themselves to give birth to a perfectly whole offspring. Some can reproduce while they’re still larvae. Though jellies lack eyes, Haddock and his colleagues have discovered proteins that comb jellies use to sense light. Comparative biologists like to joke that on the eighth day, God created comb jellies.
Comb jellies are gelatinous like jellyfish, but the similarity ends there. In body plan, jellyfish resemble the largely sessile, almost plantlike sea anemones, corals and other cnidarians: a group that dates back at least 550 million years. While jellyfish and other cnidarians have nerve cells that form a loose network in their bodies, comb jellies have a more sophisticated nervous system with a rudimentary brain and cellular connections called synapses that are also found in flies, humans and most other animals.
Yet, detailed looks at the genomes of two species of comb jellies suggest, surprisingly, that they are the more primitive animals, and not the jellyfish, sea anemones or corals, as has long been thought. It’s even possible that the sophisticated comb jelly lineage may have evolved before the brainless, gutless, muscle-less sea sponges.
With just 150 known species, comb jellies represent only a small slice of the animal kingdom. But their strangeness, right down to their DNA, threatens much of what scientists thought they knew about the origins and early evolution of animals.
The notion that comb jellies may elbow out sponges from the base of the animal tree of life is a radical one. If true, it means that comb jellies evolved nerves, muscles and other complex features, which in some ways resemble our own, independent of the ancestor that led to most animals.
Alternatively, and even more difficult for biologists to accept, is the possibility that the last common ancestor of all animals might have possessed complex features that remained in the comb jellies but were lost without a trace in the sponges, jellyfish and their kin. Both options muddle traditional assumptions that multifaceted features do not pop up and vanish willy-nilly over evolutionary time.
Leonid Moroz, a neurobiologist at the University of Florida’s Whitney Laboratory for Marine Bioscience in St. Augustine, recently sequenced the genome of the sea gooseberry comb jelly, Pleurobrachia bachei. The new genetic findings imply that “there may be many ways to make a complex animal,” he says.
Others disagree. “It would be remarkably fascinating if comb jellies evolved neurons and muscles independently, and astonishing if they were at the base of the tree,” says Graham Budd, a paleontologist at Uppsala University in Sweden. “It is effectively saying animals evolved twice. Frankly, I’m not ready to believe it.”
Tree of life — as a sapling
Long before Charles Darwin described evolution as descent with modification, zoologists noticed that animals shared certain structures. For example, humans, birds, lizards and fish all have a backbone composed of several bony vertebrae that protect a spinal cord. Rather than independently evolving this complicated structure along each of their evolutionary branches, Darwin suggested that these animals inherited a backbone from a common ancestor, which was modified over time in different lineages. That is why some snakes have 400 vertebrae and humans have 33.
When scientists draw evolutionary trees, they compare and contrast traits for clues on how animals are related. In general, biologists favor the simplest solution — usually the one in which most lineages radiating out from a common ancestor share most of the ancestor’s features. This concept of simplicity, called parsimony, has long guided thinking on animal origins.
All animals alive today descended from a clump of cells that were able to communicate and adhere to one another more than 800 million years ago. This event appears to have happened once, as did other milestones in animal evolution such as the organization of cells into tissue layers, says Claus Nielsen, a biologist at the Natural History Museum of Denmark in Copenhagen and the author of the textbook Animal Evolution.
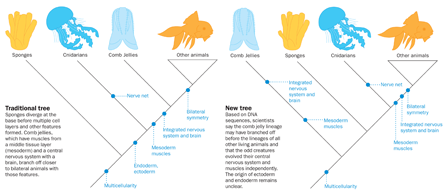
In traditional trees of life, the sponges branch off first, as multicellular animals without much specialization. Jellyfish, sea anemones and corals are thought to come later, from an ancestor with multiple cell types, and some cells organized into an outer layer of tissue surrounding the body and an inner tissue layer lining the gut. An animal with all these features plus nerve cells, a rudimentary brain and a middle tissue layer that forms muscles is traditionally thought to have given rise to comb jellies and the rest of the animals.
With the earliest animal lineages arranged in this order, major transitions paved the way for further innovations. This is evident not only in body structures that look alike, but in shared molecular underpinnings. In the case of multicellularity, many of the same proteins stick cells to one another and communicate messages between cells in all living animals. The same concept holds true for muscles and the central nervous system, which consist of several distinct parts built by networks of proteins encoded by genes. The fact that many of the interacting components are shared by all animals leaves Nielsen and many others resistant to the idea that comb jellies originated the parts on their own and then converged on a common design. “The more complicated a shared structure, the less likely it is to be convergent,” or to have evolved independently, says Nielsen. “One cannot exclude the possibility of convergence, but there is a big difference between possible and probable.”
In the 1990s, biologists predicted that studies of animal genomes would mirror the gradual addition of anatomical complexity in early animal evolution. Where humans have about 22,000 genes in their genome, it was expected that sponges, sea anemones and comb jellies would have far fewer. Yet in 2007, biologists were taken aback by a report in Science showing that the starlet sea anemone has nearly as many genes as a human. The genetic potential for complexity, it seemed, existed early on.
Comb jellies made a splash a year later. An evolutionary tree built according to similarities in select stretches of DNA, rather than shared anatomical traits, placed the comb jellies below the brainless sponges. At the time, scientists largely dismissed the finding, calling it a result of imperfect tree-building algorithms. In fact, the team initially left the finding out of its paper. “But the reviewers wanted us to say something, so we noted the result and said it needed further analysis,” says Andreas Hejnol, a coauthor on the 2008 report in Nature and an evolutionary developmental biologist at the Sars International Centre for Marine Molecular Biology in Bergen, Norway. “But privately among ourselves, we talked about what it would mean if [comb jellies] are at the base,” Hejnol says. “It would mean that they evolved complexity independently, or that the sponges lost a massive amount of complexity.”
Finding convergence
To explore that question and others, a group of biologists decided to tackle an entire comb jelly genome. They chose a walnut-shaped jelly, Mnemiopsis leidyi, which could be collected easily off the coast of Cape Cod, Mass., and reared in the lab.
In January, Andy Baxevanis, a lead investigator on the M. leidyi genome project and a comparative biologist at the National Human Genome Research Institute in Bethesda, Md., presented his team’s results in San Francisco, Calif., at the annual meeting of the Society for Integrative and Comparative Biology. According to one mathematical model that compared thousands of portions of the comb jelly’s genome to those of other organisms, comb jellies belong at the base of the animal tree of life. However, another analysis placed comb jellies below jellyfish and their kin, but it could not resolve whether sponges or comb jellies came first.
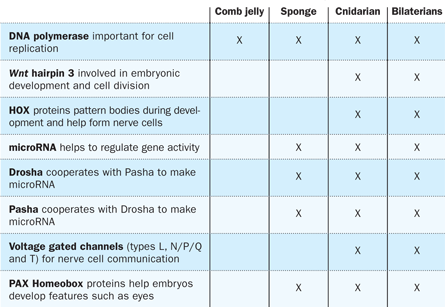
Baxevanis’ team scanned the comb jelly genome for some of the most widespread and fundamental gene families in the animal kingdom. Animal cells communicate and adhere to one another with molecules called the LIM proteins, for example, but comb jellies have fewer genes encoding these proteins than other animals. Molecular components of signaling pathways involved in cell growth and metabolism are missing in comb jellies and sponges. The Hox genes, key to early development and responsible for signaling where the brain, limbs or other body parts should form, are also absent in comb jellies and sponges. And comb jellies may be the only animals that lack both gene-regulating molecules called microRNAs as well as the molecular machinery to create them.
Baxevanis says the simplest explanation for the missing genes is that they evolved after comb jellies branched off from the ancestors of other animals, arguing for an earlier spot on the tree for the jellies.
More support for the dramatic repositioning of the comb jelly emerged at the San Francisco meeting, where in addition to Baxevanis’ talk there were 13 other presentations and posters related to the jelly genomes. Most important were new data from the group led by Moroz, which had just finished analyzing the genome of the gooseberry comb jelly — P. bachei,a symmetrical beauty with eight combs of iridescent cilia and two long sticky tentacles. In his team’s new tree of animal evolution, comb jellies also diverge at the bottommost branch, below the sponges.
Moroz, a neurobiologist, was prompted to study comb jellies out of his interest in the origin of the nervous system. He had suspected that the one in comb jellies could represent a very early form of our own.
Like Baxevanis, Moroz and his colleagues found a slew of genetic differences between the gooseberry comb jelly and the other types of animals they compared it with.
Moroz lingered on the findings that both comb jellies and sponges lack multiple genes thought to be crucial to a functioning nervous system. The result makes sense for sponges because they have no nerves, he says, but for comb jellies, “that’s shocking because they have a brain, a nervous system and complex reactions.” Comb jellies are active: Some species chase down prey and others cast out their tentacles like fishing nets. Yet neither team located genes encoding serotonin, dopamine and most other classic neurotransmitters that send messages between neurons in other animals. Absent too are proteins that, in other animals, guide the growth of neurons.
Also not present in the comb jellies’ genomes were the usual lineup of genes associated with muscles in other animals. And the muscle genes that were present in comb jellies appeared to function in unusual ways. For instance, genes that in other animals form the middle tissue layer (from which muscles arise) turned on in nerve cells in comb jellies.
Parallel evolution
Comb jellies clearly have muscles, nerve cells and a rudimentary brain. So jellies may just use a distinct set of genes to build these parts. Or the familiar genes are lurking but have mutated so much that they are unrecognizable. At the moment, no one knows which genes underlie comb jellies’ muscles and nervous systems because scientists don’t know what to look for.
The uniqueness of the comb jellies’ muscles and nervous systems on a genetic level makes the proposition that comb jellies evolved these features independently sound less preposterous. If Moroz’s assertion that comb jellies “developed complex animal innovations in parallel with other animal lineages” is true, the creatures might have started simple when they arose more than 550 million years ago. Furthermore, if the ancestor of the comb jellies was simple, sponges and the jellyfish group need not have lost complexity, even if comb jellies represent the oldest living lineage.
It’s impossible to determine what comb jellies looked like originally because gelatinous animals leave hardly any mark on the fossil record. However, paleontologists who study the Ediacaran Period (635 to 542 million years ago) say life in this era, before most modern animal lineages arose, was seriously strange. Shaped like wrinkled lips, bubbly ferns and squashed, spiral galaxies, many Ediacaran fossils cannot be neatly placed in any modern-day category. “In a sense, the Ediacaran biota may be failed experiments in animal multicellularity,” explains Douglas Erwin, a paleontologist at the Smithsonian Museum of Natural History in Washington, D.C.
Could comb jellies be the only survivors of an otherwise extinguished dynasty dating back to the Ediacaran? “I would entertain the idea,” Erwin says. “Maybe they are the sole extant representatives of what was an attempt to make something fast and predatory with an early genetic toolkit,” he speculates. In contrast, the sponges may have shared a similar toolkit but laid low. With around 8,000 species of sponges alive today, that strategy evidently worked.
Despite the evidence, many biologists are reluctant to accept that muscles and the central nervous system evolved more than once, although most accept the idea that these features have been highly modified over evolutionary time. Joseph Ryan, an evolutionary biologist at the Sars lab and a member of the M. leidyi team, attributes this conviction to human bias: “People are convinced that our nervous system is the greatest thing in the world, so they ask ‘how could it happen twice?’ ”
Understanding the true evolutionary tree would do more than shed light on animal relationships. It would also reveal whether convergent evolution is more common than biologists have assumed. If the comb jelly lineage branches off at the bottom of the tree, parsimony suggests that the comb jellies independently gained complex features. Muscles and an integrated nervous system would evolve once along their branch, and also in an animal that evolved after the sponges and the group containing jellyfish split off. The alternative — that an animal ancestor had all these features — means the features were lost once in sponges and again in the jellyfish group. Sponges, both living and fossilized, show no sign of ever having these features or the tissue layers that would be required to build them.
The fact that the comb jelly lineage landed in different places depending on the analyses Baxevanis’ team ran highlights the difficulty of reconstructing the single, true tree of life. Despite the genomes, the scientists are not there yet. But a better view of the earliest branches on the tree may not be far off. Techniques used to analyze species relationships improve every year, and new gene data from more species should help.
Studies into how comb jellies develop and operate their body parts might also reveal differences overlooked in the past when the common origin — and therefore the presumed similarity — of the muscles and nervous system was taken for granted. Mark Martindale, an evolutionary biologist at the Whitney Marine Laboratory in Florida who works on the M. leidyi project, points out that differences between animals might turn out to be beneficial.
Perhaps the proteins that help a comb jelly to regenerate its brain, nerves and muscles could reveal a potential to do the same in humans, he says. The comb jelly genome also contains elements that cause cancer in mammals, such as a notorious gene called Myc that leads to unfettered cell growth. Maybe the jellies have discovered ways to keep cancer-causing genes in check. “The bright side of all of this convergence is that by studying it comparatively, you can come up with new remedies for old problems,” Martindale says.
In December 2012, Baxevanis’ team posted the M. leidyi comb jelly’s annotated genome online, and while both teams have revealed their findings at meetings neither has published its magnum opus: the definitive comb jelly genome manuscript announcing its more ancient origin and its independent evolution of complexity.
The challenges the teams will face when they do publish are clear. But Moroz has some advice: In Darwin’s time, the theological argument was that complex systems couldn’t evolve without a creator, he says. Now, the dogma is that complexity can evolve, but not often. “It’s like we are brainwashed about complexity,” he says. If more biologists would only devote their attention to the comb jelly, he argues, they’d learn how innovative evolution can be.
Amy Maxmen is a freelance science writer in New York City.






