A chaperone protein (bottom, yellow) called SecB guides the folding of another protein (transparent) in this artist’s illustration. “Interactions with chaperones are very common for all proteins,” says Sander Tans of the FOM Institute for Atomic and Molecular Physics in Amsterdam, and they exert a strong influence on protein shape and function.
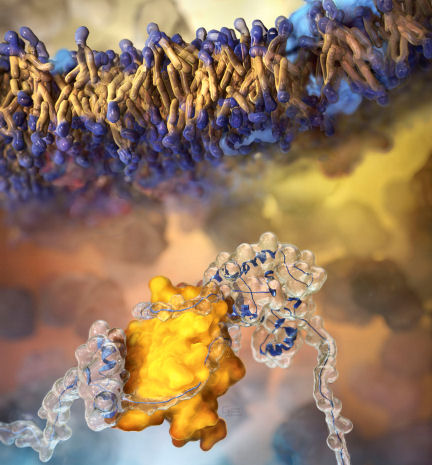
But previous studies of the molecular details of protein folding have left chaperones out of the picture. Tans and his colleagues studied the folding of maltose binding protein (MBP). By using lasers to pull apart the ends of the protein, the researchers could measure how the protein slowly unfolded and refolded, both with the chaperone present and without. The influence of the chaperone prevented MBP from folding into its final, bulky shape. Instead, it remained in a more stretched-out shape that allows the protein to pass through the cell membrane (top), the team reports in the Nov. 30 Science.





