Globin Family Grows: Blood-protein relative is in all tissues
The blood protein hemoglobin and its relative, myoglobin, carry and store life-giving oxygen in many animals. Researchers long thought these complex proteins, with their unique fold, were the only two globins in vertebrates. Other globins had been discovered in invertebrates, plants, bacteria, and fungi.
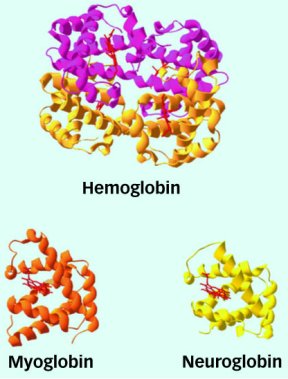
Two years ago, however, a third vertebrate globin was discovered in the human brain and named neuroglobin. Now, scientists have chanced across a fourth globin, and they say it’s present in all body cells.
Because of their high concentrations in blood and muscle, hemoglobin and myoglobin are among the best-studied proteins.
Hemoglobin-packed blood cells act as a molecular supply caravan, taking up oxygen in the lungs and then dropping it off in other tissues. The protein is responsible for blood’s ruby-red color. Myoglobin molecules reside in muscle cells, where they store oxygen for use during rigorous muscular activity.
Biologists have yet to determine the function of neuroglobin, which was discovered by Thorsten Burmester’s team at Johannes Gutenberg University in Mainz, Germany.
Even as that mystery lingers, Burmester’s team describes its discovery of the globin it calls cytoglobin in the April Molecular Biology and Evolution. At Iowa State University at Ames, another group independently announces the same discovery in an upcoming Journal of Biological Chemistry, although this team calls the molecule histoglobin.
The authors of both papers discovered the gene for the new globin by using computers to search public databases of human DNA sequences created during the human genome project. Using the technique known as gene mining, the researchers instructed computers to find DNA sequences similar to those in the genes for other globins.
The function of the new globin remains unknown, but Burmester suggests it could have a role in oxygen transport and storage similar to myoglobin’s.
Both research teams also propose that the newly discovered globin may act as an enzyme that mops up dangerous free radical molecules, such as nitric oxide, that are produced during normal chemical reactions. Nitric oxide is a signaling agent within the body, but it can wreak havoc if it’s not kept under control, says Mark S. Hargrove, a coauthor of the Iowa State study. “Bacteria have globins specifically for this purpose” and not for oxygen transport, he adds.
“If I was a betting man, I’d put my money on them both [neuroglobin and cytoglobin/histoglobin] being nitric oxide scavengers,” comments Robert Hill, who studies plant globins at the University of Manitoba in Winnipeg. Hill says he’s about to publish data suggesting that both these vertebrate globins share structural properties with several plant globins that are nitric oxide scavengers.
“Mining [the human-genome database] for genes requires a little bit of tricky programming,” says William Royer Jr., a biochemist at the University of Massachusetts in Worcester. However, “it’s becoming an increasingly common technique.” Royer says he believes that the same method could yield discoveries of further vertebrate globins.