Gut Check
The bacteria in your intestines are welcome guests
New York and London are famous for both their congestion and the diverse origins of their residents. But if you’re looking for the ultimate teeming metropolis of immigrants, check out the large intestine. In people, some 500 to 1,000 kinds of bacteria reside in this part of the gastrointestinal (GI) tract, and these gut microbes outnumber all the cells in your body, perhaps by as much as a factor of 10.

|
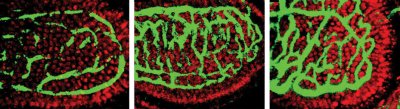
BACTERIAL BOOST. The intestinal blood vessel system (green) of a germfree mouse (left) is less mature than that of mice colonized by typical gut microbes (middle) or Bacteriodes thetaiotaomicron alone (right). |
“The density of this society is mind-boggling,” says Jeffrey I. Gordon of Washington University School of Medicine in St. Louis.
It’s a society overlooked by most microbiologists, who generally stick to the myriad bacteria that cause disease. Yet some scientists argue that it’s shortsighted to ignore what they call the microflora living in our intestines.
“What these bacteria do definitely makes a very significant contribution to our health–or lack thereof,” says Mark Schell of the University of Georgia in Athens, who studies an intestinal microbe called Bifodobacterium longum.
Shell and a few other researchers have recently begun to probe exactly what individual microbes do for or to the intestine.
Consider Bacteriodes thetaiotaomicron. Although not as well known, it’s more than 1,000 times as abundant in the guts of people and mice as the extensively studied bacterium Escherichia coli. Some researchers have proposed that in return for a steady food supply, B. thetaiotaomicron breaks down indigestible complex carbohydrates into easily absorbed sugars and produces other substances, such as vitamins, that benefit its host.
There may be much more to this microbe-host relationship, however. About a decade ago, Gordon chose B. thetaiotaomicron as a prototypical germ for studying how microbes influence the GI tract. This bacterium normally becomes a predominant member of the intestinal community about the time an animal is weaned from its mother’s milk. Gordon’s research team has discovered that the microbe can turn on specific intestinal genes, promote the growth of blood vessels necessary for the gut’s function, and trigger production of a chemical that may kill competing bacteria. Investigators are now asking just how much gut bacteria regulate the developing and adult human body.
“Bacteria do an awful lot for us and with us,” says Gordon. “Most people’s views of bacteria are of menacing, disease-promoting entities. Au contraire, I think that most of our encounters with bacteria are mutually beneficial, friendly, and part of our normal biology. . . . They’ve insinuated themselves into our biology and coevolved with us.”
Sweet-talking germ
Perhaps the best way to understand the significance of intestinal microorganisms is to see what happens when an animal doesn’t have them. During the past 50 years, researchers have created germfree mice and rats by delivering the animals by cesarean section into sterile environments and maintaining them there. “It’s a very demanding technology,” says Gordon. Scientists have generally used such germfree animals to study how particular pathogens cause diseases.
One of the most striking aspects of a germfree rodent is that it must consume about 30 percent more calories to maintain its body weight than a typical rodent does. Germfree animals are also unusually susceptible to infections, presumably because the microflora in a normal gut ward off foreign pathogens.
As a way to study animals hosting a simplified society of gut bacteria, Gordon and his colleagues have introduced B. thetaiotaomicron into germfree mice. Their first significant discovery was that the bacterium could change what sugars the intestine makes.
The surfaces of intestinal cells of typical mice are coated with complex sugars containing the simple sugar fucose and B. thetaiotaomicron consumes the fucose for energy. In germfree mice, however, fucose production ceases around the time of weaning.
If B. thetaiotaomicron colonizes a germfree mouse before weaning, however, normal fucose synthesis continues throughout life, the researchers found. Through a still undiscovered signal, the microbe apparently induces the intestinal cells to make one of its favorite foods. The bacterium even has a fucose sensor that informs it when this food source is scarce, according to Gordon and his colleagues.
The capacity of B. thetaiotaomicron to instruct intestinal cells to make fucose was just a hint of things to come. To get a more comprehensive picture of the bacterium’s influence, Gordon’s group turned to microchip-size devices, called DNA microarrays, that monitor the activity of thousands of genes at once (SN: 3/8/97, p. 144).
With such instruments, the scientists took a snapshot of the gene activity in the mouse intestine. By comparing tissue from germfree mice and mice hosting B. thetaiotaomicron, the team found that the presence of the bacterium significantly reduces or boosts the activity of about 100 of the approximately 25,000 rodent genes in the microarray survey.
Some of the intestinal genes triggered by the microbe help mammals absorb and metabolize sugars and fats, Gordon and his colleagues reported in 2001. Other activated genes fortify the cellular barrier that prevents bacteria, both dangerous and friendly, from sneaking out of the intestine into other tissues and the bloodstream. And yet other affected genes determine how the intestine detoxifies compounds and how the gut matures.
“We were amazed at the breadth of normal intestinal functions affected by a single microbe,” says study coauthor Lora V. Hopper. Gordon adds, “It’s difficult to anticipate the full range of host functions that might be manipulated by these microbes.”
The genetic activity that the researchers didn’t see in the bacteria-colonized mice was interesting, too. Even though the originally germfree mice had never encountered B. thetaiotaomicron before, there was no increase in activity of the genes underlying an immune or inflammatory response. That’s a reflection of the microbe’s still mysterious skill at convincing a host that it’s a friendly visitor and not a danger, says Gordon.
Raising fences
Among the intestinal genes activated by B. thetaiotaomicron is one suspected to stimulate the growth of new blood vessels. The finding spurred Gordon’s group to investigate the microbe’s control over the system of blood vessels that runs through the GI tract. These blood vessels are crucial to a body’s absorption of nutrients.
The researchers discovered that their germfree mice have a poorly formed network of the capillaries that normally supply the inner intestinal surface with its blood supply. This could partly explain the difficulty that germfree animals have absorbing nutrients.
The team also found that it could stimulate germfree mice to grow a normal network of intestinal capillaries by exposing the animals to either a full complement of microflora or just B. thetaiotaomicron. The investigators reported the finding in the Nov. 26, 2002 Proceedings of the National Academy of Sciences.
This is a vivid illustration that the physical development of the gut can depend on the microbes that normally inhabit animals, says Gordon. The researchers also found cells in the mouse gut that seems to work with the microbes to spur vessel growth.
In the small intestine, so-called Paneth cells normally secrete antimicrobial compounds (SN: 8/26/00, p. 135: Available to subscribers at Tales from the crypts: Cells battle germs). This keeps the intestine healthy by protecting other cells that continually replenish the gut lining. Gordon’s team created germfree versions of mutant mice that lack Paneth cells and found that B. thetaiotaomicron couldn’t trigger the maturation of blood vessels in such rodents. While most investigators have regarded Paneth cells simply as defenders against invading bacteria, it makes sense that these cells mediate interactions between a host and its natural microflora, says Gordon. “What better cell to respond to the presence or absence of a microbe?” he remarks.
The Paneth cell is at the heart of another microbe-intestine interaction uncovered recently by Gordon’s group. One of the intestinal genes triggered in germfree mice by B. thetaiotaomicron encodes a protein called angiogenin 4 or Ang4. Cancer researchers are particularly interested in this protein, because they have evidence that it nourishes tumors by creating new blood vessels. Gordon’s team suspected that Ang4 plays a role in the intestinal blood vessel maturation that they had documented earlier. Indeed, it turned out that Paneth cells make Ang4 and secrete it when they detect bacteria.
While the suspicion that Ang4 makes intestinal blood vessels has not yet been proven, it looks like the protein has a more certain role. It can kill several bacteria and fungi that cause diseases in mammals, Gordon, Hooper, and their colleagues report in the March Nature Immunology. In contrast, B. thetaiotaomicron and other common residents of the mouse intestines are largely resistant to Ang4.
“One interpretation of the interaction between host defense and the resident flora is that the resident bacteria that are resistant to Paneth-cell secretions stimulate these host-defense mechanisms to prevent competition by nonresident bacteria. The host in turn benefits by decreasing its exposure to potential pathogens,” says Tomas Ganz of the University of California, Los Angeles in a commentary accompanying the March report.
Hooper agrees that the normal inhabitants of the gut may use Paneth cells and Ang4 to raise what she calls an “electric fence” to keep out competing microbes. Beyond fending off foreign pathogens, such fences may also keep typical intestinal microbes within the gut. “Anything can become a pathogen if it crosses the fence,” she says.
Eating leftovers
Scientists have estimated that the hundreds of bacterial species within the human gut may together possess as many unique genes as a person does, and perhaps far more. “How much of our biology is dependent on metabolic traits encoded in the collective genomes of our microbial partners?” asks Gordon.
Investigators have begun to address that question. For example, Schell recently worked with scientists at the Nestlé Research Center in Lausanne, Switzerland, to unravel some of the genetic secrets of B. longum. This microbe typically colonizes the intestines of a newborn mammal, thrives during the breast-feeding period, and then subsides after weaning, when B. thetaiotaomicron and other bacteria take hold. Nestlé incorporates B. longum into some of its products, such as infant formulas and yogurts, to promote gastrointestinal health.
In the Oct. 29, 2002 Proceedings of the National Academy of Sciences, Schell and his colleagues unveiled the entire DNA sequence of B. longum and identified a large roster of genes for enzymes that break apart sugars and other edible substances. Some of these enzymes may degrade complex sugars found in breast milk, speculates Schell. Others, such as ones that apparently break down plant gums, may help the bacterium survive later in its host’s life when B. longum is in the minority in the intestines.
The bacterium appears to break down substances that B. thetaiotaomicron and other Bacteriodes can’t handle. “It seems to be more specialized for the leftovers of metabolism,” says Schell. In a strategy similar to Gordon’s, investigators at Nestlé are now using germfree mice to evaluate B. longum‘s impact on intestinal genes.
Gordon’s team is drawing its own insights from the group’s recent deciphering of B. thetaiotaomicron‘s genome. Among that microbe’s nearly 4,800 genes, several hundred encode proteins that bind carbohydrates, enzymes that degrade bonds between sugars, or enzymes that create new sugars, the investigators reported in the March 28 Science. And the activity of many of these genes appears to be regulated by genes encoding molecules related to known environmental sensors, suggesting that the microbe can monitor the contents of the intestines and quickly deploy the molecular machinery needed for it to digest nutrients.
“This organism has a sweet tooth. It knows how to process carbohydrates,” says Gordon.
Over the next 5 to 10 years, predicts Schell, researchers will decode the genomes of many more intestinal microbes. Investigators may also begin to address such issues as whether a person’s diet changes his or her intestinal microflora. “I think the gut population of a vegetarian is clearly going to be different” from that of a meat eater, says Schell.
Gordon offers an even more provocative question: Do intestinal microbes influence a person’s weight? “Over time, could relatively minor differences in the ability to extract nutrients in some individuals predispose them to obesity?” he asks.
The complicated nation of bacteria within our intestines is a “window into our biology and how we’ve evolved as a species,” concludes Gordon.
****************
If you have a comment on this article that you would like considered for publication in Science News, send it to editors@sciencenews.org. Please include your name and location.
To subscribe to Science News (print), go to https://www.kable.com/pub/scnw/
subServices.asp.
To sign up for the free weekly e-LETTER from Science News, go to http://www.sciencenews.org/subscribe_form.asp.