Laser light rewrites memories in mice
Nerve cells in the hippocampus help the brain modify recollections

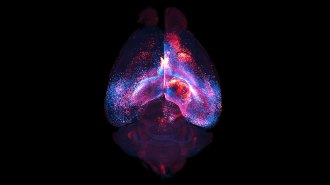
ALTERED Mice’s bad memories turned into good ones and vice versa with a flash of light. The switch occurred when nerve cells (red) storing the memory were activated while a mouse experienced something new. The black region (center) shows the location of the light-delivering optical fiber.
R. Redondo
With a burst of light, scientists can change good mouse memories into bad ones, and bad ones into good. The results, published August 27 in Nature, underscore how memories are not written in stone, and bring scientists closer to understanding how nerve cells in the brain create and store memories.
The study provides a “much more precise handle on some of the steps of memory formation than we’ve had before,” says neuroscientist Richard Morris of the University of Edinburgh. One day, such knowledge may lead to treatments for people who struggle with unwanted negative memories.
The results are the latest in the effort to manipulate mouse memories with optogenetics, a technique that uses light to control specific nerve cells in the brain. In earlier experiments, scientists made mice afraid of a harmless room, essentially layering fearful memories onto previously neutral experiences (SN: 8/24/13, p. 18; SN: 4/21/12, p. 10).
In the new work, MIT scientists led by Susumu Tonegawa go further by transforming a once-negative memory into a pleasant one and a once-positive memory into a bad one. The team exposed male mice to either of two distinct situations: Some received small electric shocks while others spent time with females. A molecular trick marked neurons that stored the memory of each experience so that the cells, and the memory they evoke, could be later activated with laser light piped through tiny optical fibers embedded in the mice’s brains. The targeted neurons were located in the hippocampus or the amygdala, two brain structures involved in memory.
After tagging these memories, the researchers called them back up with light. In a completely new environment, the mice chose whether to have the laser stimulate their brain — a decision that allowed the scientists to see if the stimulation, and thus the memory, was pleasant or not. Mice with foot-shock memories spent less time getting stimulated, while mice that had positive memories of spending time in the company of a female relished the laser, the team found.
In the next step, the team changed the positive or negative experience of a memory by calling up the memory with light while each mouse experienced the opposite situation, says study coauthor Roger Redondo. For example, the researchers used light to evoke the memory of a shock while the animal was in the company of females, causing the negative memory to be diluted with a more positive one.
After the memory switch, the mice were again put in a different environment. Those that had their pleasant memories sullied stimulated themselves less with the laser. Meanwhile, mice that had their nasty memories made more pleasant went for the laser more often. And back in the room where they had received shocks, these mice acted less frightened, spending less time frozen in fear and more time sniffing, a sign that they were looking for a female.
The memory switch seems to be driven by cells in the hippocampus, not in the amygdala, the team found.
Emotional memory switching isn’t a new concept, says Redondo. The same thing happens when a person experiences a terrible meal at a formerly beloved restaurant. And a similar process might underlie exposure therapy for post-traumatic stress disorder, in which a person relives a traumatic experience in the hopes of forming new, less upsetting associations. What makes the new technique different is that it switched memories quickly and without returning to the scene of the event, he says.
“We know the circuit so well that we can go inside the brain, tweak the circuit and achieve something that otherwise we’d need drugs or weeks and hours of behavioral therapy to get,” says Redondo.
Tonegawa points out, however, that the technology doesn’t exist to enable a similar switch in people. “It’s not something we can do next week or next year,” he says. But he’s hopeful that the research might ultimately help people with traumatic memories.