Lessons from the Torpid
Hibernators have some helpful tips for keeping humans healthy
On February 2, groundhog weatherman Punxsutawney Phil roused from hibernation to predict six more weeks of winter. Scientists may snicker at people who think they can learn about the arrival of spring from a furry rodent, but researchers aren’t laughing when it comes to learning about human health from animals that check out for the winter.


Understanding how hibernators, including ground squirrels, marmots and bears, survive their long winter’s naps may one day offer solutions for problems such as heart disease, osteoporosis and muscular dystrophy.
Despite appearances, hibernation is not the same as going to sleep for a long time. It is extreme living by any measure. For about half the year, hibernating animals stay in their dens or burrows in a state of suspended animation, waking up every now and again to go to the bathroom. Most hibernators eat or drink nothing, living solely off the fat they built up before winter began.
To make fat stores last, animals lower their metabolism and body temperatures. Black bear body temperatures drop to about 33º Celsius (about 91º Fahrenheit), but the bodies of most small mammal hibernators, such as ground squirrels and woodchucks, plunge to nearly freezing. Some Arctic ground squirrels hold steady at subzero temperatures. For all these animals, heartbeats and breathing nearly cease. These are feats of physiological daring that non-hibernators, including humans, could never survive.
Yet sometimes humans do have to deal with more moderate versions of hibernators’ challenging circumstances — following periods of weight gain, immobilization or blood loss, for example. So, many scientists think some tricks of the hibernation trade might be a boon to human medicine.
Trying to find ways to treat human diseases with the help of animals is nothing new. Researchers often attempt to solve medical riddles by first creating versions of disorders, such as muscular dystrophy or stroke, in mice or other lab animals and then figuring out what goes wrong. But even when researchers know what’s broken, a fix is not always obvious. Hibernators, though, have already found ways to cope with body and lifestyle changes that would lead to disease in humans.
In one effort to tap into hibernators’ solutions, Ole Fröbert, a cardiologist at –rebro University Hospital in Sweden, and his colleagues are investigating the body transformations that Scandinavian brown bears undergo each winter, including how they have such high cholesterol but no increased risk for heart disease. “We see the brown bear as a living library of information,” Fröbert says. “It’s an animal that has solved a multitude of problems that humans face.”
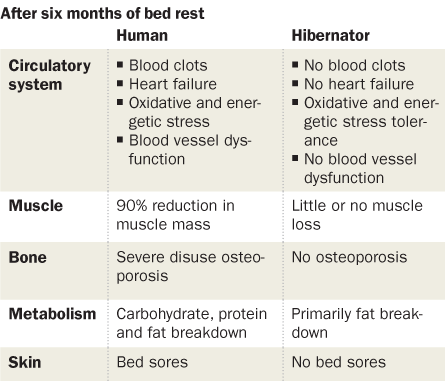
Insights gleaned from bears and ground squirrels may also help researchers prevent or reverse bone and muscle loss in space travelers, elderly people and children with muscular dystrophy. Such lessons could reveal ways to render people confined to bed rest immune to blood clots, bed sores and muscle and bone atrophy. Therapies designed to mimic ground squirrel hibernation strategies could save soldiers and accident victims from bleeding to death. And these critters just might hold secrets for reducing damage from strokes or heart attacks, and even for preserving organs for transplant after they are cut off from a blood supply and put on ice.
Extra pounds, no problem
Nearly everything about the way an animal’s body works changes when it hibernates, and preparations start weeks or months in advance. The first order of business is to fatten up.
“Fat is where it’s at for a hibernator,” says Matthew Andrews, a molecular biologist at the University of Minnesota Duluth who studies 13-lined ground squirrels. “You bring your own lunch with you.” Packing lunch is necessary because the animals go on the world’s strictest diet during the winter, surviving entirely off their white fat. “They have their last supper in October; they don’t eat again until March,” Andrews says.
Bigger fat stores mean a greater chance of surviving until spring. “If they go in really chunky, nice and roly-poly, that’s going to be a good hibernator,” he says.
Bears also watch their waistlines expand in the months before settling in for the season. The brown bears Fröbert studies pack on the pounds by chowing down on up to 40 kilograms of blueberries a day. Such gluttony among humans could have severe consequences: Obesity is associated with a greater risk of heart disease and diabetes, among other ailments.
To see how fattening up affects Scandinavian brown bears, Fröbert and his colleagues ventured into the wilds of Sweden following signals given off by radio transmitters or GPS devices on tagged bears.
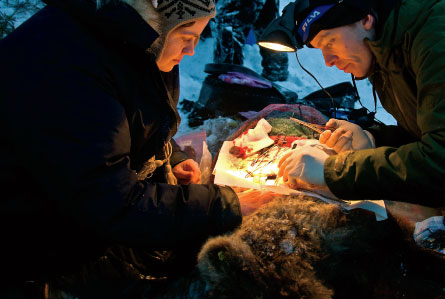
Bears can be dangerous close-up. Even hibernating bears can rouse to action quickly, so scientists tracking down bears in the winter use darts to tranquilize the animals from a distance. Scientists studying the bears in the summer tranquilize them from a helicopter.
Once a bear is under the tranquilizer’s influence (which takes about five minutes), the scientists have 60 minutes max to get the animal from its den, weigh and measure it, draw blood samples and do minor surgeries to collect fat and other tissues. The bear is returned to its den by minute 61.
Precious materials collected during this high-pressure encounter need to be analyzed within 24 hours, so the researchers often test for levels of cholesterol or certain proteins in the blood while working in the snow or at a nearby research station. A pilot sometimes flies samples from field sites to a lab in Denmark in order to meet the deadline, Fröbert says. Samples such as bones and arteries that can’t be collected from live bears come from bears killed by hunters during the legal hunting season.
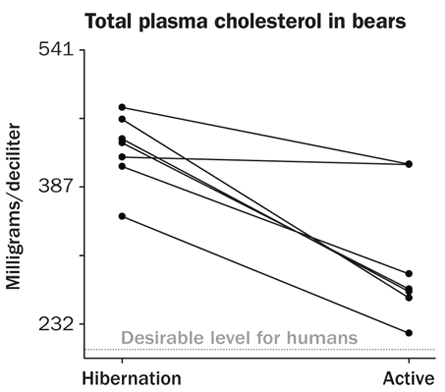
Recent analyses revealed that Scandinavian brown bears spend the summer with blood cholesterol levels considered high for humans; those values then increase substantially for hibernation, Fröbert and his colleagues reported online January 10 in Clinical and Translational Science. These “very, very fat” bears with high cholesterol also get zero exercise during hibernation. Lolling about in the den pinches off blood vessels, contributing to sluggish circulation. “That cocktail would not be advisable in humans,” Fröbert says. It’s a recipe for hardened arteries, putting people at risk for heart attacks and strokes.
Even healthy young adult humans can develop fatty streaks in their arteries that make the blood vessels less flexible, but the bears don’t build up such artery-hardening streaks. “Our bears, they had nothing,” Fröbert says. It’s not yet clear how the bears keep their arteries flexible, but Fröbert hopes to find some protective molecule that could stave off hardened arteries in humans as well.
Sturdy skeletons
The bears’ cholesterol-defying arteries are just one of the evolutionary tricks allowing hibernators to spend six months as furry couch potatoes without negative side effects.
Such inactivity wreaks havoc on the human body, which can’t maintain strong muscles and skeletons without weight-bearing physical activity. People normally lose a little bone as they age; about 1 to 2 percent of the minerals in bone begin to leach out of women’s spines and hips each year after menopause. But people on bed rest or in the weightlessness of space may lose 3 to 4 percent of their hip bone minerals each month, says endocrinologist Peter Vestergaard of Aarhus University Hospital in Denmark. Bed sores and blood clots also plague the sedentary human. And yet, come spring, hibernators emerge free of clots and sores and with their skeletons and muscles intact.
Rita Seger ventures into the backwoods of Maine to learn how American black bears keep their bones healthy during hibernation. She and her colleagues from the Maine Department of Inland Fisheries and Wildlife track bears right to their dens, thanks to radio collars fitted on the bears in the spring. The trips usually involve packing a portable centrifuge and an X-ray machine powered by the battery from a cordless drill.
Comparing X-rays of the paws of tranquilized hibernating bears with X-rays taken of paws from bears that had been killed by hunters revealed no differences, suggesting hibernation doesn’t melt bone the way bed rest does for humans. Seger and colleagues also compared blood samples from the hibernating bears with blood drawn from bears that were active in the spring, looking for chemical clues to how the skeletons are maintained.
Seger, a general internal medical doctor and physiologist at the University of Maine in Orono, and her team found proteins in the bears’ blood indicating that bone-building cells are less active in hibernating animals than in spring bears, reporting the results in the December Bone. Humans on bed rest also build less bone than normal, but bone is torn down at a greater rate, making for an overall loss. The black bears balance slower bone building with a reduction in bone loss, the researchers revealed, keeping the skeleton strong.
Some of the bears’ balancing skills may be transferable. Already, black bear parathyroid hormone, which helps determine how much bone is built or reabsorbed, is in development as a possible treatment for human osteoporosis, says Seth Donahue, a biomedical engineer at Colorado State University in Ft. Collins. Donahue is working with a biotechnology company to test the hormone’s bone-sparing power in rats. When researchers remove the ovaries from female rats to simulate menopause, the rats’ bones become spongy. But rats treated with the bear parathyroid hormone retain more bone, preliminary studies show.
Bears may also hold the secret to avoiding blood clots, such as those that develop when people sit still too long on plane trips, Fröbert says. He and his colleagues measured the blood-clotting activity of cells called platelets taken from humans and from brown bears shortly after the bears emerged from their dens in the spring. The bears’ platelets were about half as active as the human platelets, the researchers reported in 2010 in Thrombosis Journal. Fröbert’s team is trying to find out why the bear cells have lower clotting power and how that power changes during hibernation.
It’s in their blood
Bears may have a lot of helpful information to impart, but they are not easy to work with. Some scientists have turned to a smaller hibernating critter that can be studied in the lab, the 13-lined ground squirrel. This rat-sized creature may reveal a strategy to help with another blood problem: massive blood loss.
On a visit to Ronald Cohn’s lab at Johns Hopkins University Medical School in early December, most of the 13-lined ground squirrels were curled into drowsy, striped balls, their faces tucked into the cream-colored fur on their bellies. One slumbering squirrel’s sides rose and then fell with a single breath. About 20 seconds later, the squirrel breathed again.
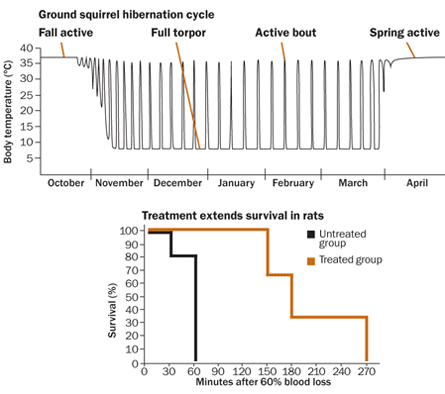
Once these squirrels go into the refrigerator for the winter, they will enter full torpor, as scientists call deep hibernation. The animals will take only two to four breaths and their hearts will beat just two to four times per minute, Cohn says.
“The theory has been that hibernators are exquisitely adapted to function in the cold,” says Sandra Martin, an evolutionary geneticist at the University of Colorado School of Medicine in Aurora. “We don’t find that. Our results suggest that they’re exquisitely adapted to shut everything down in the cold.… It’s almost as if the animal is putting itself in an ice bucket, slowing everything down to save energy.”
But about every two weeks, the hibernators do something that makes little energetic sense. They warm up and move around, just for a bit.
“It’s expensive to be up at high temperatures in the cold,” says Hannah Carey, a physiologist at the University of Wisconsin–Madison School of Veterinary Medicine. While the animals are up and about — about 10 to 12 hours for ground squirrels — they may urinate and move around, but generally do not eat or drink. Bears go the entire winter without excreting anything at all, though they too stir occasionally.
The 13-lined ground squirrels hibernating in Cohn’s refrigerator go from dormant to alert within minutes. “They will all be as active as this guy,” Cohn says, tapping a gloved finger on the cage of a bright-eyed ground squirrel, sending it darting for cover. It’s soon out front again, whistling curiously at Cohn and his company. After a few hours, the animal will curl into a torpid ball with no regard for Cohn, who is trying to understand how the ground squirrels maintain muscles during torpor.
The ground squirrels’ up-and-down cycles cause repeated loss and restoration of blood flow to parts of the animals’ bodies, a situation most humans face only when something has gone horribly wrong.
In people, blood clots, heart attacks, strokes, accidents or even sitting or lying too long in one position may shut off blood supply to cells that need it. Oxygen deprivation from reduced blood supply can damage tissues and organs. But restoring blood flow comes with its own problems, Carey says. The influx of oxygen-rich blood causes little power plants called mitochondria to go wild churning out molecular energy for the cell in which they reside. By-products of the energy-producing reactions — molecules called oxidants or oxygen radicals — can damage the proteins, DNA and fats that make up cells. If the damage is bad enough, the cells will die and may contribute to tissue damage or organ failure.
Ground squirrels are susceptible to this type of damage in the summer, but come winter, hibernating squirrels become impervious to the effects of oxygen deprivation and restored blood flow.
To find out how the squirrels handle the constant near-freezing and thawing, Andrews and his colleagues compared gene activity in summer ground squirrels with activity in hibernating animals, during full torpor and bouts of activity.
The team found that hibernating ground squirrels keep their bodies going during the temperature drop by fueling the organs with breakdown products from fat, rather than glucose as summer ground squirrels do. During brief active bouts, levels of a chemical called melatonin shoot up in the ground squirrels’ blood. Melatonin is known mainly as a hormone that helps regulate the body’s daily rhythms, but it is also a powerful antioxidant — just what ground squirrels need to fight off damage from oxygen radicals as their blood begins to surge.
A cocktail including fat breakdown products (substances called ketone bodies) and melatonin might help people who have lost a lot of blood, such as soldiers with battlefield injuries or accident victims, hold on until they can get a transfusion, Andrews reasoned. So his team tested concentrated solutions of the cocktail in rats. Untreated, rats that lost 60 percent of their blood died within an hour. But the cocktail extended rats’ survival to three hours or more, Andrews and colleagues reported in 2010 in Shock. The therapy is now being tested in pigs, and may soon enter human clinical trials.
If the cocktail ends up saving lives, ground squirrels and the power of evolution that has given them extreme survival skills will deserve the glory. “I keep telling people over and over again that the credit goes to the basic hibernator biology,” Andrews says. “We never would have come up with that combination on our own.”
Further research into how hibernators ride out the winter may yield other therapeutic solutions for humans. Even though people don’t go into torpor, they may have the molecular equipment to pull off some milder hibernation-linked feats that could boost health during hard times. “I would argue that the hardware is mostly there,” Martin says. “It is just a matter of learning how to use it.”