Nanomaker’s Toolkit
Scientists harness charge, magnetism and even DNA to guide matter’s assembly into new materials
When assembling a jigsaw puzzle, just shaking up the box and dumping the pieces in a pile probably isn’t the best strategy. The pieces won’t fit themselves together by chance. But in the nanoworld, this approach could prove surprisingly fruitful.
It might take the fun out of doing the puzzle, but scientists are now figuring out how to make the pieces move, on their own, into the desired positions for creating new materials. In this case, the puzzle pieces are nanoparticles, tiny collections of atoms smaller than one ten-thousandth of a millimeter across. Properly guided, these particles could assemble into materials useful as conductors or catalysts, with the potential for making smaller computer circuits, better biosensors and more efficient solar panels.
Because nanoparticles are small, a large proportion of their atoms are near the particle’s surface. Having fewer neighbors, those relatively unconfined atoms can link in unusual ways, giving materials made of nanoparticles novel properties.
But the same characteristic that makes nanostructures useful —size — also makes working with them no small task. Engineering on the nanoscale is like building a ship in a bottle while wearing mittens.
It would be far cheaper and easier, researchers agree, if nanoparticles could just arrange themselves into nanomaterials —like dropping the pieces of the ship into the bottle and then sitting back to watch the ship build itself. What scientists are working on now is finding the right chemistry — creating just the right conditions so that natural properties such as charge or magnetism direct the pieces of the ship to come together just so, with the mast above the deck and never below or to the side.
This idea, called self-assembly, isn’t exactly new. Examples range from the simple separation of oil and vinegar in a bottle of salad dressing to the complex movements of proteins and enzymes — themselves nanosized — reacting in living cells. Scientists have long been inspired by these naturally self-assembling systems. But designing self-assembling systems in the lab, with nanoparticles, presents its own scale of difficulty. And making self-assembled nanomaterials grow large enough to actually be useful is even more challenging.
Previous work on self-assembly set nanoparticles organizing according to size, shape and charge. But the outcomes were not as specific as desired. “It’s like trying to build a house by randomly throwing bricks together,” says physicist Alexei Tkachenko of the University of Michigan in Ann Arbor. “You get a crude version of a house, but it doesn’t look very pretty.”
Using different techniques, independent groups of scientists have recently figured out better ways to make the nanoparticles go where they are supposed to go. One group of researchers uses magnetic forces to position particles. Another has figured out how to use DNA to herd nanoparticles into exact conformations. And others can make nanoparticles spread themselves into a thin layer over a surface, changing the properties of the material beneath.
Charged to get together
Before self-assembly, scientists used other tricks for making nanoscale impressions. But the tricks have limits.
One technique for making something in the nanoscale is to shrink photolithography, where light serves as a tool for etching intricate circuits onto the tiniest computer chips. A lens focuses ultraviolet light through a stencil to create a pattern on a light-reactive material. This patterned material then stamps its pattern onto a silicon film.
The technique works well — but just setting up to form one specific structure is expensive. And the technique imprints only the surface of the material, making 2-D nanostructures but not 3-D ones such as those required for solar panels.
While still in its early days, the technology of self-assembling systems of nanoparticles is close to building useful, complex, even 3-D materials, researchers believe. “We have some challenges to overcome,” says physicist Oleg Gang of the Center for Functional Nanomaterials at the Brookhaven National Laboratory in Upton, N.Y. But “we have the potential to make complicated systems just by mixing up some components.”
Some scientists are using nature’s self-assembling systems as models for designing their own. “We’re trying to mimic nature, where everything is self-organized,” Gang explains.
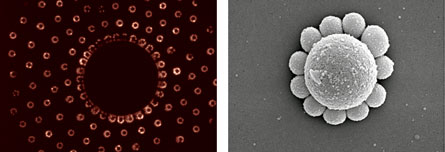
For physicist Elena Shevchenko of Argonne National Laboratory in Illinois, inspiration comes from the structure of opal gemstones. Opals are a naturally occurring nanomaterial, made up of silica nanoparticles that self-assemble into a crystal structure. Shevchenko and her colleagues aimed to build, or “grow,” similar crystals from nanoparticles of various materials in the lab.
“Nanoparticles are not identical or uniform in size and shape,” says bioengineer John Crocker of the University of Pennsylvania in Philadelphia. “We can take for granted that atoms will crystallize in a certain way. But we don’t fully understand how nanoparticles will act in a certain liquid and how they will sit together.”
Shevchenko and her colleagues figured out that when certain types of nanoparticles are suspended in liquid, natural electric charges among the particles make them assemble into crystal structures. The scientists figured out how to combine not just one type of nanoparticle but two different types to form the opal-like structures, called binary superlattices. The sizes and concentrations of the nanoparticles determined how they packed together to form crystals.
Evaporating the liquid stabilized the structure, the researchers reported in Nature in 2006, when Shevchenko was with Columbia University and IBM. The choice of particles and liquid steered the self-assembly.
The process successfully created 2-D films of nanocrystals, but adding more types of nanoparticles could allow scientists to precisely tailor the material, and even add a dimension. An alloy of two metals, for example, has properties of both. Similarly, a material made from two or three types of nanoparticles would carry extra properties.
Of course, the same charges that form the nanocrystals also limit the choices for possible structures. The attractive and repulsive forces steered the nanoparticles into only one conformation, but the researchers think that other combinations could be more useful.
So some researchers have come up with a new message about designing useful self-assembling systems, and they’re saying it with flowers.

Magnetic beats electric
A team at Duke University in Durham, N.C., created a swirling broth of iron nanoparticles and larger polystyrene beads bouncing around randomly. An applied magnetic field stops the swirling and acts on the iron nanoparticles. Magnetized, the iron helps steer polystyrene beads to line themselves up. Under a microscope, researchers can watch as the polystyrene particles arrange themselves into flower structures.
Looking down the microscope at the tiny flowers floating in solution “was beyond my wildest imagination,” says materials scientist Benjamin Yellen of the Magnetic Nanosystems Group at Duke, who worked on the project. “It wasn’t just that the flowers formed, but how reliably they formed. It just turned out that this system worked.”
Yellen and his colleagues were the first to apply magnetic forces and manipulate interactions among particles in a liquid. Polystyrene beads embedded with iron could, with help from the iron nanoparticles, follow the magnetic field. The nonmagnetic beads lined up differently. This shift, coupled with interactions among the particles, moved the beads into the flower shapes, the team reported in the Feb. 19 Nature.
“The magnetic fields moved the nanoparticles the way we wanted them to move regardless of the charge on the particle,” Yellen says.
The polystyrene particles were microsized, large compared with the tiny nanoparticles that Shevchenko and her team steered into structures. But theoretical work suggests that smaller particles can also be manipulated using magnetic forces and similar broths. The technique would allow nanoparticles to form groups called nanoclusters — in this case, nanoclusters of three or more different types of nanoparticles.
Guiding particles into clusters is a way to make materials that have exciting new properties. “It’s like making atoms connect to make molecules,” Gang explains. Nanoclusters have different properties than their nanoparticles, just as water differs from hydrogen and oxygen.
Follow that DNA
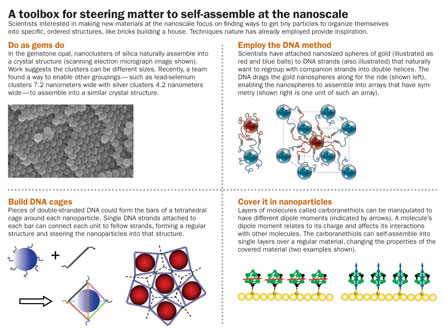
Some researchers have come up with a different strategy to form nanoclusters: using DNA and its system for holding two strands together. “The idea is to use DNA as a smart glue to hold nanoparticles together,” Crocker says.
The DNA used for self-assembly is synthetic and does not code for any genes. But these random strands of DNA do have important properties learned from biology. The two strands of DNA in the familiar double helix structure bind together because of attractive forces between the chemical “letters” in the DNA code. Unzipping the helix leaves each strand of DNA open to attach to one nanoparticle, so that each nanoparticle has a DNA tail. Mixed in a liquid, the matching DNA strands bind together again, dragging the nanoparticles with them.
Such DNA shepherds could help in designing complex self-assembling systems, those containing many different components, the researchers say. “We can assign names to the different pieces of the structure. We can tell A to interact with B but not with C,” says Gang, a coauthor of several papers on DNA-assisted self-assembly. This level of control could enable the formation of intricately patterned nanomaterials from several different types of nanoparticles.
Making changes in the DNA code that defines how the strands link could make the interactions among nanoparticles stronger or weaker. “It’s a recognition system that was developed by nature,” Gang says.
Gang’s team managed to get nanoparticles to come together but didn’t have much control over how the particles organized. So Gang and his colleagues next used DNA to link nanoparticles to a solid surface. Once the nanoparticle is held in place, it’s easier to control how it interacts and connects with other nanoparticles, the researchers report in the May Nature Materials.
As part of earlier work, Gang’s team had optimized the DNA-nanoparticle linkages. Previous researchers had found it difficult to control how many pieces of DNA would attach to each nanoparticle, or exactly where on the nanoparticle the DNA would attach. Some nanoparticles could have two DNA arms, and others could have 10. So instead of forming regular, ordered structures, the nanoparticles stuck together in random clumps.
Reporting in a 2008 Nature paper, Gang and colleagues described a technique that links nanoparticles into 3-D crystals. Attaching longer pieces of DNA to spherical nanoparticles of gold helped to make regular crystal structures, the researchers reported. The long DNA pieces wrap around the nanoparticle, enveloping it. Since the DNA surrounds the particle uniformly, the randomness of the DNA attachment doesn’t matter.
Although the researchers managed to make small nanoclusters and nanoparticle crystals self-assemble, making larger materials has been “surprisingly difficult,” Crocker says. When the crystals assemble, they are floating in water. “We don’t want squishy, wet materials,” Crocker says, “but when we dry the structure out, the conformation [of the nanoparticles] changes.” Filling the gaps between the nanoparticles with a gel or ceramic could be a way to make a solid material, the researchers say.
In the future, researchers hope to be able to use DNA to build cages around nanoparticles, and use the cages as building blocks for a self-a