Ominous signals: Genes may identify the worst breast cancers
Some women with breast cancer respond well to treatment while others succumb to the disease, even when the cancer in both cases appears to have been caught early and was treated similarly. A growing pool of evidence suggests that the genetic nuances of tumor cells account for the contrasting outcomes.
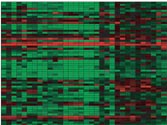
To turn those genetic traits to medical advantage, several research groups have begun scanning DNA in breast-tumor cells to determine which of thousands of genes are most or least active in aggressive cancers.
A U.S.-Dutch team doing such gene profiling reports in the Jan. 31 Nature that certain patterns of activity crop up more frequently in the most deadly breast cancers–those that spread beyond the breast–than in cancers that remain in remission after initial treatment.
At present, physicians derive a breast cancer prognosis from the tumors size, the extent of its spread, the degree to which the tumor cells differ from normal cells, molecular characteristics of the tumor cells, and the patients age. Tumor removal and radiation therapy cure most women whose cancer is confined to the breast. Nonetheless, one-fourth of such women subsequently have cancer crop up elsewhere, says Stephen P. Ethier, a molecular biologist at the University of Michigan School of Medicine in Ann Arbor.
To obtain genetic clues to this risk, the researchers analyzed samples of 78 breast tumors that had been surgically removed from women. All the patients had breast cancer that was confined to the breast at the time of diagnosis. Of these, 34 women had cancer arise outside the breast within 5 years.
Using microarray analysis–a lab technology that reveals activity in individual genes–the researchers scanned roughly 25,000 genes in the tumor cells. Of the genes, 4,968 showed up as either especially busy or sluggish in at least three tumors.
Next, the researchers used a computer to look for patterns of gene overactivity or underactivity associated with aggressive tumors. Assessed this way, 70 genes stood out. The team tested the accuracy of this genetic profile by using it to determine whether a tumor in a new set of samples came from a woman who had an aggressive cancer or had one that remained under control.
The profile enabled the scientists to predict correctly–albeit retrospectively–the fate of 17 of 19 cancer patients, says study coauthor Stephen H. Friend, a molecular biologist at Rosetta Inpharmatics in Kirkland, Wash.
The emerging technology is enabling scientists to think differently about how we classify breast cancer, says Ethier. While the technique still doesnt permit a physician to give all women accurate prognoses, a refined version could ultimately . . . change the way a person might be treated, he says.
The next step for researchers will be to test the method on hundreds of additional patients, Friend says.
Especially good candidates would be women whose breast cancer is caught and removed early and who dont appear to need chemotherapy. Microarray analysis could turn up a dangerous pattern of gene activity in some of those women, pinpointing who should get further treatment, such as chemotherapy, he says.
Although such a genetic profile is currently very expensive to generate, Friend says the cost is dropping and could be down to hundreds of dollars per test in the near future.
Meanwhile, the 70 genes highlighted in this study include many that hadnt been previously implicated in breast cancer. Researchers might start investigating these genes individually to see if any presents a target for therapy, Friend says. The genes that are overactive in aggressive cancers and the proteins they encode may offer new avenues to slow breast cancer growth.







