It started with droughts in Australia and Ukraine. Wheat yields dropped and countries clamped down on grain exports. With a hungry biofuel market also gobbling up corn, the cost of food soared.




Sensing an opportunity for profit, farmers sought more fertilizer to nourish their fields. But high oil prices had increased the cost of processing phosphate rock, which provides a key ingredient in fertilizer. With rising demand and tight supply, phosphate rock prices leaped from about $45 per metric ton to $80, then $135, then $367 — a roughly 700 percent spike in just one year. As food prices continued to surge, riots erupted from Haiti to Burkina Faso.
The crisis, which unfolded from 2007 to 2008, foreshadows what might happen more regularly if the world runs short of phosphate rock, the main source of phosphorus for fertilizer. Phosphorus, an essential nutrient for life, helps power the growth of crops. By adding the element to fields, farmers boost their yields and churn out the billions of tons of food needed to feed Earth’s growing population.
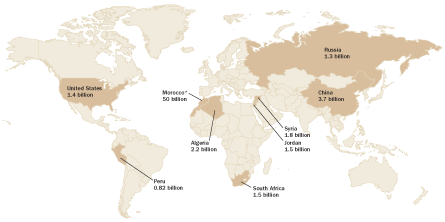
But the planet holds a limited amount of easily accessible phosphate rock. Just a few countries, such as Morocco, China and Algeria, control large deposits that can be mined with existing technology at a reasonable cost. The United States is currently a major supplier, but its reserves are dwindling. As high-quality deposits become depleted, remaining untapped reserves generally contain less phosphorus. Some researchers predict that the annual amount of phosphorus retrieved from mined phosphate rock could peak sometime this century.
And how much is left isn’t the only concern. Political disputes over ownership of the Western Sahara territory — which along with Morocco contains about three-quarters of the world’s remaining known reserves — raise fears that geopolitical instability could threaten the global phosphorus fertilizer supply.
Aware of the growing risk, researchers are investigating how phosphorus in soil and waste can be conserved and recovered. A frequently cited 2009 study in Global Environmental Change shows that people waste a flabbergasting amount of this precious resource: Each year, about 80 percent of phosphorus mined for fertilizer is washed off farms, left in livestock manure, dumped into landfills or otherwise squandered before the food even reaches people’s mouths. Many farmers still overfertilize their fields, despite initiatives to reduce fertilizer use.
To combat these losses, some researchers are engineering plants that suck up leftover phosphorus from the soil, while others are trying to pull phosphorus from wastewater, using everything from nanoparticles to algae.
Through these efforts, scientists and engineers are drawing attention to a neglected problem. Potential fossil fuel and water shortages regularly make headlines, but phosphorus often takes a backseat. “I call it the biggest problem we’ve never heard of,” says James Elser, an ecologist and co-organizer of the Sustainable Phosphorus Initiative at Arizona State University in Tempe.
Lazy plants
Life needs phosphorus. It’s part of DNA’s backbone and contributes to the structure of cell membranes. “Life without phosphorus would be like life without oxygen or carbon or hydrogen,” says Dana Cordell, a sustainability researcher at the University of Technology Sydney and cofounder of the Global Phosphorus Research Initiative. “Life would not exist.”
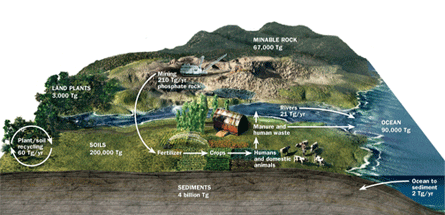
Phosphorus exists primarily in sediments and rock. When marine organisms die or are eaten and excreted by other animals, some of the phosphorus in their bodies sinks to the ocean floor and gets buried in sediment, eventually becoming rock. Over millions of years, that rock shifts and lifts back up to Earth’s surface. The rocks weather and break down, forming new soil containing phosphorus that can nourish plant growth. Phosphorus from dead plants and the carcasses or excrement of animals also enters the ground.
Farmers face two big problems in getting to that phosphorus. First, the weathering process is very slow. Second, after phosphorus enters the soil — whether from rock weathering or land animals — much of it ends up bound to iron, aluminum or calcium, which prevent plant roots from absorbing it. So, many soils don’t contain enough accessible phosphorus to support vigorous crops.
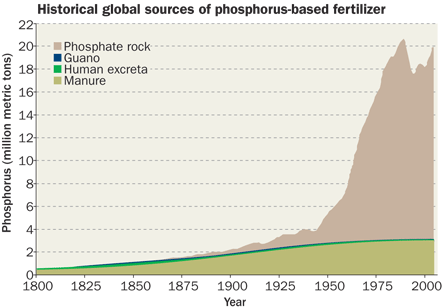
For centuries, people improved crop yields by fertilizing their farms with phosphorus-rich waste such as manure and bat droppings. They didn’t always realize they were using phosphorus, but they knew those materials made plants grow better. In the 1900s, companies started ramping up production of chemical fertilizer containing phosphorus, helping trigger a worldwide explosion in farm productivity.
Lavished with a rich supply of fertilizer, domesticated crop plants weren’t under any pressure to scrape together phosphorus from nutrient-poor soils. So their roots became lazy, says Roberto Gaxiola, a molecular biologist at Arizona State University and an investigator with the Sustainable Phosphorus Initiative. “The original plants used to grow with whatever they had,” he says. “And then we started spoiling them.”
In one effort to recover phosphorus, Gaxiola is now coaxing plants to slurp up more phosphorus from the earth. If Gaxiola can get plants to grow bigger roots, they would release more protons, which would acidify the soil. Extra acid frees some phosphorus into a form that plants can take up.
In a 2007 study in Plant Biotechnology Journal, Gaxiola’s team genetically engineered mouse-ear cress plants to make extra copies of a protein called AVP1. The researchers knew that this protein makes plants grow bigger root systems, though they didn’t yet know why. Under low-phosphorus conditions, the engineered plants sprouted more and bigger leaves than unmodified plants, the team found.
In another experiment, the researchers found a likely cause for the improved growth: Engineered plants grown in the low-phosphorus nutrient mixture turned the mixture more acidic, which would free up more phosphorus.
Next, the team tried similar experiments in crop plants. Tomato plants that made AVP1 could produce more fruit in low-phosphorus soil than unmodified tomatoes could, outweighing them by 82 percent. Engineered rice grew taller, and shoots weighed 50 percent more.
Gaxiola suspects the AVP1 protein helps the plant channel more sugar from the leaves to the roots, allowing a larger root system to flourish. These harder-working crops could recover phosphorus stuck in soil that has been heavily fertilized, he says. For the last two years, Gaxiola’s team has grown lettuce in previously fertilized fields and found that the engineered plants perform better than unmodified plants do, even with little or no added fertilizer. Ultimately, engineered plants might mean less fertilizer use from the start.
The research is promising, says Sigrid Heuer, a molecular biologist at the Australian Centre for Plant Functional Genomics in Glen Osmond. But she notes that strict regulations on genetically engineered crops may hinder the approval of transgenic plants. Recent work by Heuer and her colleagues while she was at the International Rice Research Institute in Los Ba±os, the Philippines, used traditional breeding methods to create rice varieties that can tolerate low-phosphorus conditions.
In a paper published in Nature in August, Heuer’s team showed that a gene called PSTOL1, found in a traditional rice plant variety that had been exposed to little fertilizer, could make modern rice plants grow more grain in phosphorus-deficient soil. The team’s new rice varieties containing this gene produce 20 percent higher yields in the field. The researchers hope to make one available to farmers in two to three years.
Down the drain
Another major leak in the phosphorus cycle is human waste. Once crops are harvested, they end up on dinner plates or feed livestock later eaten by humans. Since people generally get more phosphorus than they need, they excrete almost all of what they consume. “There’s a whole bunch of that going down the drain in the form of wastewater,” says Andrew Shilton, an environmental engineer at Massey University in Palmerston North, New Zealand.
Wastewater treatment plants do remove phosphorus from sewage. In the 1960s, people became concerned that phosphorus-laden wastewater released into lakes was triggering the growth of algal blooms. Bacteria consuming the dead algae suck up oxygen, suffocating other aquatic life. To reduce phosphorus levels in wastewater, many treatment plants now add chemicals such as aluminum sulfate, which bind the phosphorus and settle out as sludge. Some treatment plants also use bacteria to gobble up the phosphorus.
But most treatment plants don’t recover phosphorus in an easily reusable form. Chemical sludge is often thrown into a landfill. Although treatment plants use processes that digest phosphorus-stuffed bacteria and offer the remains as fertilizer, transporting the bulky, dilute slurry to individual farms is expensive.
One promising strategy is to harvest phosphorus from wastewater as part of a mineral called magnesium ammonium phosphate, or struvite. Struvite forms naturally when phosphate, magnesium and ammonium mix in the right proportions. Wastewater treatment plant operators have generally considered struvite a nuisance because it builds up as concrete-like gunk in magnesium-lined pipes.
But struvite can also be made in a more controllable way. In the early 2000s, researchers at the University of British Columbia in Vancouver built roughly cone-shaped reactors that create enough turbulence in wastewater to trigger the formation and growth of struvite crystals. “Once that crystal forms, it’s almost unbreakable,” says team leader Don Mavinic, a wastewater treatment and quality researcher. With a little added magnesium, the reactors could produce 98 percent pure, pearl-like struvite pellets, the researchers reported in Water Science & Technology in 2008.
That struvite can then serve as fertilizer. A company called Ostara Nutrient Recovery Technologies Inc. in Vancouver now uses the technology pioneered by Mavinic’s team to produce struvite fertilizer pellets, named Crystal Green, from wastewater. Ostara has installed its reactors at wastewater plants in Oregon, Virginia and Pennsylvania and is adding reactors to plants in Wisconsin, Saskatchewan and the United Kingdom. Wastewater plants typically call on bacteria to mop up phosphorus first, then digest the microbes to release the phosphorus into a liquid stream that gets channelled to Ostara’s reactors. The reactors can recover about 85 to 90 percent of the phosphorus that flows into them, says Phillip Abrary, CEO of Ostara. And the pellets are small and concentrated, making them easy to transport to farms.
Another phosphorus recovery technique is inspired by methods for removing arsenic from drinking water. Ben Martin, a chemical wastewater engineer at Cranfield University in England, came across a product consisting of small resin beads embedded with iron oxide nanoparticles. When water passes through a column containing the beads, the nanoparticles tightly bind to arsenic-containing molecules.
Since arsenic and phosphorus have similar chemical properties, this material could also bind phosphate, a molecule made of one phosphorus and four oxygen atoms that’s commonly found in wastewater.
To find out how well the product removed phosphorus, Martin pumped synthetic, wastewater-mimicking solutions into columns containing the beads. The material captured an average of 98.7 percent of the phosphorus, Martin reported in his 2010 doctoral thesis. He could later release phosphorus from the beads by adding sodium hydroxide.
Other researchers have found that melter slag, a waste product of steel-making that contains iron oxide, also can remove phosphorus from wastewater. But Martin’s approach has the advantage that nanoparticles collectively have a high surface area and can thus bind a lot of phosphorus.
Over the next four years, Martin plans to test the nanoparticle process at a wastewater plant in England. He also wants to investigate whether different types of nanoparticles release phosphorus more easily during the recovery step.
Closing the cycle
Phosphorus is in demand not just for food crops, but also for biofuel production. Algae, for example, produce and store oil in their cells that could be used for biofuel. But growing large amounts of algae requires fertilizer, including phosphorus.
Jonathan Trent, a marine biologist at NASA Ames Research Center in Moffett Field, Calif., wanted to come up with a way to grow algae without competing with farms for land, water and fertilizer. Instead of jockeying for the same resources as farmers, Trent thought, why not take the phosphorus and other nutrients from wastewater that coastal cities dump into the ocean? The water could be redirected into floating reactors off the coast to help algae grow. Later, the algae’s oil could be harvested to make biofuel. Phosphorus from other parts of the algae, such as their DNA, might also be recovered to produce fertilizer.
Trent’s idea led to the development of Offshore Membrane Enclosures for Growing Algae, or OMEGA, described in Biofuels in September. His team built 110-liter reactors made of long, flexible plastic tubes and tested them in outdoor tanks in Santa Cruz, Calif. Then the researchers scaled up to a 1,600-liter system in tanks of seawater at a San Francisco wastewater treatment plant. The water contained pharmaceutical and personal-care products such as shampoo, but these didn’t hinder algae from growing, the team found. As for the phosphorus, the algae “absorb it almost instantly,” says Trent.
The project has drawn interest from countries around the world, including India, Australia, New Zealand, Morocco and Saudi Arabia, says Trent. But to be affordable, the infrastructure will probably need to be multipurpose. For example, seafood producers could grow mussels or oysters beneath the docks that hold the reactors. And researchers still need to find out if the reactors will stand up to storms or have a negative effect on marine ecosystems, says Shilton.
One can imagine other exotic solutions for recovering phosphorus, such as fishing it out of landfills or mining a nearby asteroid. But such strategies are likely to be expensive and difficult. They strike Elser as rather silly when there is already plenty of phosphorus that is more readily accessible. “Why don’t we just take better care of our poop?” he asks.
Even as new technologies plug some leaks in the phosphorus cycle, efforts to reduce demand could make a big difference. More farmers are finding ways to reduce fertilizer use, both by more precisely calibrating the amount they need to apply and by better controlling erosion to keep more soil, and thus phosphorus, in the fields. Eating less meat could make a difference too, since a carnivorous diet requires more phosphorus than a vegetarian one, says Cordell.
Developing countries will also need inexpensive, low-tech solutions. In Nepal, researchers have investigated cheap ways of making struvite from urine and are now expanding the work to South Africa. And a product called Peepoo allows people to collect their urine and feces in a single-use, urea-containing bag that converts the waste into sanitized fertilizer. And an international organization called Máshumus has been training Latin American farmers to extract phosphorus from animal bones.
In some ways, solving the phosphorus problem is less daunting than managing other resources. With fossil fuels, “once they’re burned, you can’t unburn them,” Elser says. But phosphorus can be reused; it’s just a matter of not letting it slip away. “Once we have a closed phosphorus cycle,” he says, “it could go on forever.”
Roberta Kwok is a freelance science writer in Burlingame, Calif.






