Scientists have long thought that the surface atoms of a solid fit together snugly and stably, like floor tiles. Now they’re finding that surface atoms of copper–and maybe other materials–roam randomly and widely within their orderly grid.
The findings, from independent teams in the Netherlands and New Mexico, may shed light on how materials form layers on other materials. An important application of such layering is the fabrication of integrated circuits and micromachines, such as tiny pressure and motion sensors.
The results also warn that future nanometer-scale components may suffer from the newfound atomic rearrangements, says Raoul van Gastel of Leiden University in the Netherlands. He and his colleagues stumbled upon the surface commotion while studying how indium impurities affect copper-crystal growth.
In the Feb. 19 Physical Review Letters, the Dutch team reports using a scanning tunneling microscope to observe the positions of indium atoms within a copper surface. The scientists scanned the same 400-atom-by-400-atom region as often as every 2 seconds.
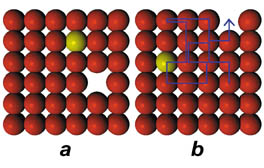
They noted that a patch of surface would remain unchanged for many scans. Then, suddenly, many indium atoms would move within the next few images. The movements suggested that atoms were hopping up and moving across the surface before dropping back in. Or, because the surfaces were warm enough, agitated indium atoms were moving into holes left in the surface layer when copper atoms occasionally hopped off the surface’s edge, van Gastel says.
“One of things that surprised us was the indium atoms made so-called long jumps” of up to five grid spaces between images, he recalls. The jumps’ timing and lengths, together with other evidence that indium atoms can’t easily enter the surface layer, pointed to leftover holes.
When an atom escapes the surface’s edge, the researchers propose, a neighboring atom fills the vacancy, causing the hole to shift by one grid space. That process repeats some 100 million times per second, shoving the vacancy around.
“You know the [toy] slide puzzles with 15 numbers and one missing? It’s the perfect analogy,” says Brian S. Swartzentruber of Sandia National Laboratories in Albuquerque. “To get numbers in the right places, you move the vacancy around.”
According to the Dutch model, a vacancy pops up every half minute or so in any particular surface patch. Each vacancy’s rapid zigzagging affects many atoms, some of them repeatedly. These multiply disturbed atoms appear to take long leaps. Nearly all copper atoms in the surface layer also shuffle around, but they’re indistinguishable from each other, so their motions can’t be tracked.
Just a few fast-moving vacancies could account for all the motion. At room temperature, “one in 6 billion atoms in the surface is missing–that’s like one person in the entire population of the Earth,” van Gastel says. Overall, the surface motion dislodges any given atom about once every 30 or 40 seconds, he says.
Using a different method, Swartzentruber says he and his coworkers at Sandia came to “basically . . . the same conclusion” as the Dutch group did. In an unpublished study, the Sandia team used a scanning tunneling microscope tip to track one palladium atom at a time within a copper surface.
Surface scientists “are very excited” about the findings, comments Karina Morgenstern of the Free University of Berlin. “We should go back . . . and see if some unexplained things might be due to this phenomenon,” she says. For instance, chemists make wide use of metal surfaces as catalysts. Morgenstern speculates that some catalytic behavior may be influenced by the newfound surface motion.






