The brain may clean out Alzheimer’s plaques during sleep
If sleep deprivation puts garbage removal on the fritz, the memory-robbing disease may develop

THE CLEAN CYCLE Lack of sleep may contribute to Alzheimer’s disease by robbing the brain of the time it needs to wash away sticky proteins.
Michael Morgenstern
Neuroscientist Barbara Bendlin studies the brain as Alzheimer’s disease develops. When she goes home, she tries to leave her work in the lab. But one recent research project has crossed into her personal life: She now takes sleep much more seriously.
Bendlin works at the University of Wisconsin–Madison, home to the Wisconsin Registry for Alzheimer’s Prevention, a study of more than 1,500 people who were ages 40 to 65 when they signed up. Members of the registry did not have symptoms of dementia when they volunteered, but more than 70 percent had a family history of Alzheimer’s disease.
Since 2001, participants have been tested regularly for memory loss and other signs of the disease, such as the presence of amyloid-beta, a protein fragment that can clump into sticky plaques in the brain. Those plaques are a hallmark of Alzheimer’s, the most common form of dementia.
Each person also fills out lengthy questionnaires about their lives in the hopes that one day the information will offer clues to the disease. Among the inquiries: How tired are you?
Some answers to the sleep questions have been eye-opening. Bendlin and her colleagues identified 98 people from the registry who recorded their sleep quality and had brain scans. Those who slept badly — measured by such things as being tired during the day — tended to have more A-beta plaques visible on brain imaging, the researchers reported in 2015 in Neurobiology of Aging.
In a different subgroup of 101 people willing to have a spinal tap, poor sleep was associated with biological markers of Alzheimer’s in the spinal fluid, Bendlin’s team reported last year in Neurology. The markers included some related to A-beta plaques, as well as inflammation and the protein tau, which appears in higher levels in the brains of people with Alzheimer’s.
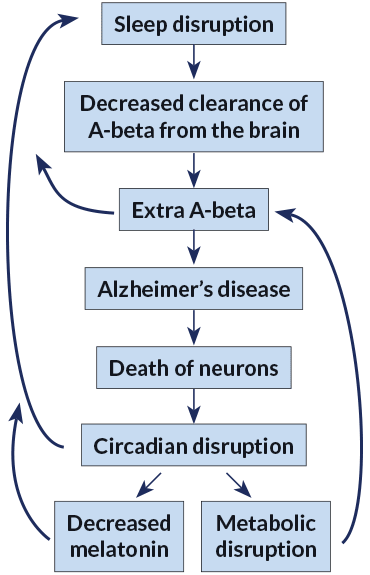
Bendlin’s studies are part of a modest but growing body of research suggesting that a sleep-deprived brain might be more vulnerable to Alzheimer’s disease. In animal studies, levels of plaque-forming A-beta plummet during sleep. Other research suggests that a snoozing brain runs the “clean cycle” to remove the day’s metabolic debris — notably A-beta — an action that might protect against the disease. Even one sleepless night appears to leave behind an excess of the troublesome protein fragment (SN Online: 7/10/17).
But while the new research is compelling, plenty of gaps remain. There’s not enough evidence yet to know the degree to which sleep might make a difference in the disease, and study results are not consistent.
A 2017 analysis combined results of 27 studies that looked at the relationship between sleep and cognitive problems, including Alzheimer’s. Overall, poor sleepers appeared to have about a 68 percent higher risk of these disorders than those who were rested, researchers reported last year in Sleep. That said, most studies have a chicken-and-egg problem. Alzheimer’s is known to cause difficulty sleeping. If Alzheimer’s both affects sleep and is affected by it, which comes first?
For now, the direction and the strength of the cause-and-effect arrow remain unclear. But approximately one-third of U.S. adults are considered sleep deprived (getting less than seven hours of sleep a night) and Alzheimer’s is expected to strike almost 14 million U.S. adults by 2050 (5.7 million have the disease today). The research has the potential to make a big difference.
Dream weavers
It would be easier to understand sleep deprivation if scientists had a better handle on sleep itself. The brain appears to use sleep to consolidate and process memories (SN: 6/11/16, p. 15) and to catalog thoughts from the day. But that can’t be all. Even the simplest animals need to sleep. Flies and worms sleep.
But mammals appear to be particularly dependent on sleep — even if some, like elephants and giraffes, hardly nod off at all (SN: 4/1/17, p. 10). If rats are forced to stay awake, they die in about a month, sometimes within days.
And the bodies and brains of mice change when they are kept awake, says neurologist David Holtzman of Washington University School of Medicine in St. Louis. In one landmark experiment, Holtzman toyed with mice’s sleep right when the animals’ brain would normally begin to clear A-beta. Compared with well-rested mice, sleep-deprived animals developed more than two times as many amyloid plaques over about a month, Holtzman says.
He thinks Alzheimer’s disease is a kind of garbage collection problem. As nerve cells, or neurons, take care of business, they tend to leave their trash lying around. They throw away A-beta, which is a leftover remnant of a larger protein that is thought to form connections between neurons in the developing brain, but whose role in adults is still being studied. The body usually clears away A-beta.
But sometimes, especially when cheated on sleep, the brain doesn’t get the chance to mop up all the A-beta that the neurons produce, according to a developing consensus. A-beta starts to collect in the small seams between cells of the brain, like litter in the gutter. If A-beta piles up too much, it can accumulate into plaques that are thought to eventually lead to other problems such as inflammation and the buildup of tau, which appears to destroy neurons and lead to Alzheimer’s disease.
About a decade ago, Holtzman wanted to know if levels of A-beta in the fluid that bathes neurons fluctuated as mice ate, exercised, slept and otherwise did what mice do. It seemed like a run-of-the-mill question. To Holtzman’s surprise, time of day mattered — a lot. A-beta levels were highest when the animals were awake but fell when the mice were sleeping (SN: 10/24/09, p. 11).
“We just stumbled across this,” Holtzman says. Still, it wasn’t clear whether the difference was related to the hour, or to sleep itself. So Holtzman and colleagues designed an experiment in which they used a drug to force mice to stay awake or fall asleep. Sure enough, the A-beta levels in the brain-bathing fluid rose and fell with sleep, regardless of the time on the clock.
A-beta levels in deeply sleeping versus wide-awake mice differed by about 25 percent. That may not sound like a dramatic drop, but over the long term, “it definitely will influence the probability [that A-beta] will aggregate to form amyloid plaques,” Holtzman says.
The study turned conventional thinking on its head: Perhaps Alzheimer’s doesn’t just make it hard to sleep. Perhaps interrupted sleep drives the development of Alzheimer’s itself.
Published in Science in 2009, the paper triggered a flood of research into sleep and Alzheimer’s. While the initial experiment found that the condition worsens the longer animals are awake, research since then has found that the reverse is true, too, at least in flies and mice.
Using fruit flies genetically programmed to mimic the neurological damage of Alzheimer’s disease, a team led by researchers at Washington University School of Medicine reversed the cognitive problems of the disease by simply forcing the flies to sleep (SN: 5/16/15, p. 13).
Researchers from Germany and Israel reported in 2015 in Nature Neuroscience that slow-wave sleep — the deep sleep that occupies the brain most during a long snooze and is thought to be involved in memory storage — was disrupted in mice that had A-beta deposits in their brains. When the mice were given low doses of a sleep-inducing drug, the animals slept more soundly and improved their memory and ability to navigate a water maze.
Gray matters
Even with these studies in lab animals indicating that loss of sleep accelerates Alzheimer’s, researchers still hesitate to say the same is true in people. There’s too little data. Human studies are harder and more complicated to do. One big hurdle: The brain changes in humans that lead to Alzheimer’s build up over decades. And you can’t do a controlled experiment in people that forces half of the study’s volunteers to endure years of sleep deprivation.
Plus the nagging chicken-and-egg problem is hard to get around, although a study published in June in JAMA Neurology tried. Researchers from the Mayo Clinic in Rochester, Minn., examined the medical records of 283 people older than 70. None had dementia when they enrolled in the Mayo Clinic Study of Aging. At the study’s start, participants answered questions about their sleep quality and received brain scans looking for plaque deposits.
People who reported excessive daytime sleepiness — a telltale sign of fitful sleep — had more plaques in their brains to start with. When checked again about two years later, those same people showed a more rapid accumulation than people who slept soundly.
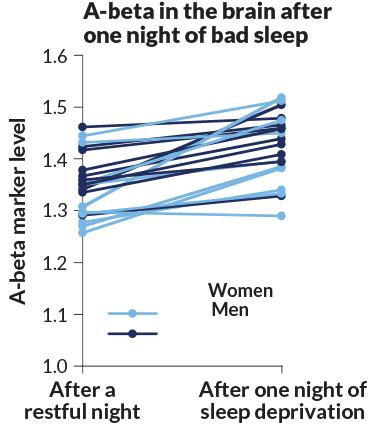
Other scientists have used brain scans to measure what happens to A-beta in people’s brains after a sleepless night. Researchers from the National Institutes of Health and colleagues completed a study involving 20 healthy people who had a brain scan while rested and then again after they were forced to stay awake for 31 hours.

Nora Volkow, head of the National Institute on Drug Abuse in Bethesda, Md., led the study. She is interested in sleep’s potential connections to dementia because people with drug addiction have massive disruptions of sleep. For the study, the researchers injected people with a compound that latches onto A-beta and makes it visible under a PET scanner.
The sleep-deprived brains showed an increase in A-beta accumulation that was about 5 percent higher in two areas of the brain that are often damaged early in Alzheimer’s: the thalamus and hippocampus. Other regions had lesser buildup.
“I was surprised that it was actually so large,” says study coauthor Ehsan Shokri-Kojori, now at the National Institute on Alcohol Abuse and Alcoholism. “Five percent from one night of sleep deprivation is far from trivial.” And while the brain can likely recover with a good night’s sleep, the question is: What happens when sleep deprivation is a pattern night after night, year after year?
“It does highlight that sleep is indispensable for proper brain function,” Volkow says. “What we have to question is what happens when you are consistently sleep deprived.” The study was published April 24 in the Proceedings of the National Academy of Sciences.
As tantalizing as studies like this may seem, there are still inconsistencies that scientists are trying to resolve. Consider a study published in May in Sleep from a team of Swedish and British researchers. They set out to measure levels of A-beta in cerebrospinal fluid and markers of neuron injury in 13 volunteers, sleep deprived and not.
The first measurements took place after five nights of sound sleep. Then participants were cut back to four hours of sleep a night, for five nights. Four participants even lasted eight days with only four hours of nightly sleep. After good sleep versus very little, the measurements did not show the expected differences.
“That was surprising,” says Henrik Zetterberg of the University Gothenburg in Sweden. Given the previous studies, including his own, “I would have expected a change.”
He notes, however, that the study participants were all healthy people in their 20s and 30s. Their youthful brains might cope with sleep deprivation more readily than those in middle age and older. But that’s just a hypothesis. “It shows why we have to do further research,” he says.
Rinse cycle
Questions could be better answered if scientists could find a mechanism to explain how sleepless nights might exacerbate Alzheimer’s. In 2013, scientists revealed an important clue.
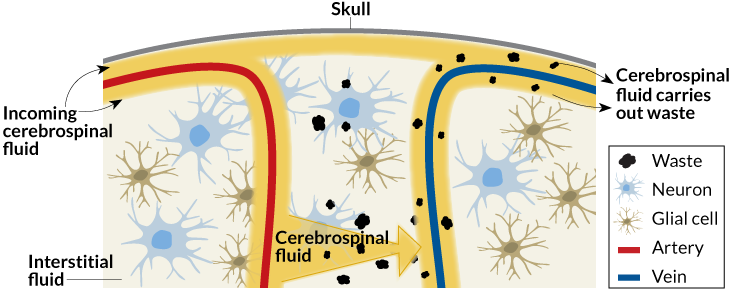
The lymphatic system flows through the body’s tissues to pick up waste and carry it away. All lymphatic vessels run to the liver, the body’s recycling plant for used proteins from each organ’s operation. But the lymphatic system doesn’t reach the brain.
“I found it weird because the brain is our most precious organ — why should it be the only organ that recycles its own proteins?” asks Maiken Nedergaard, a neuroscientist at the University of Rochester in New York. Maybe, she thought, the brain has “a hidden lymphatic system.”
Nedergaard and colleagues decided to measure cerebrospinal fluid throughout the brain. When mice were awake, there appeared to be little circulation of fluid in the brain. Then the team examined sleeping mice. “You take mice and train them to be quiet under a microscope,” Nedergaard says. “The mice after a couple of days feel very calm. Especially if you do it during the daytime when they are supposed to be sleeping, and they are warm and you give them sugar water. They’re not afraid.”
Slumbering stream
Flow of cerebrospinal fluid in a mouse’s brain is much higher during sleep (left, red) than when the animal is awake (right, green).
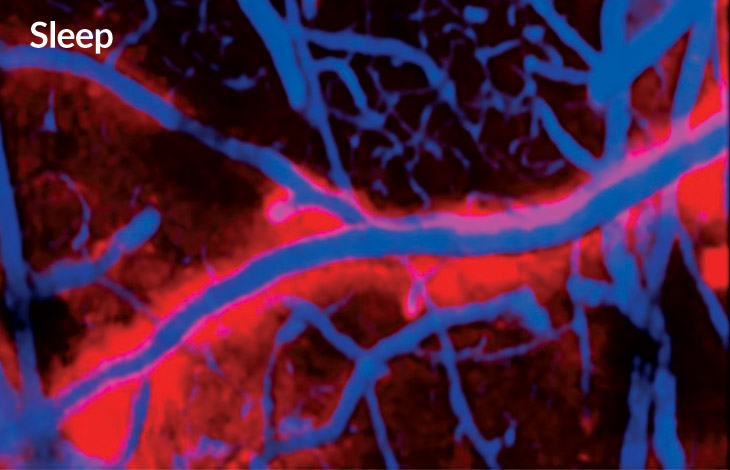
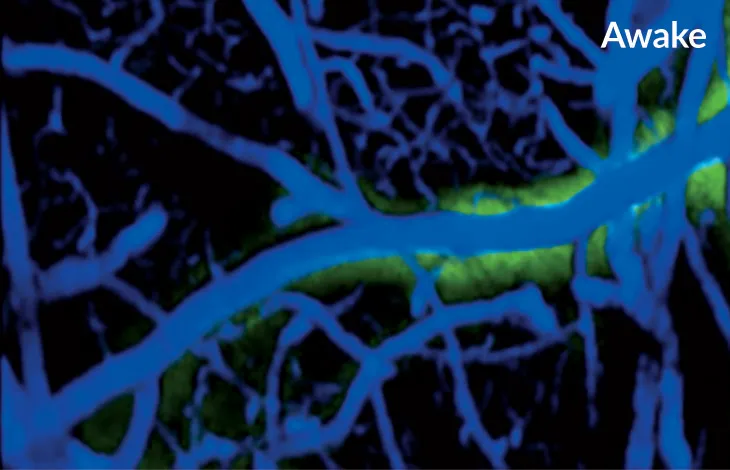
The day of the experiment, the scientists made a hole in the mice’s skulls, placed a cover over it and injected a dye to measure cerebrospinal fluid in the brain. During sleep, the spaces between the brain cells widened by about 60 percent and allowed more fluid to wash through, taking the metabolic debris, including A-beta, with it.
“It’s like the dishwasher turned on,” Nedergaard says. She named this phenomenon the “glymphatic system” because it appears to be controlled by glial cells, brain cells that help insulate neurons and perform much of the brain’s routine maintenance work (SN: 8/22/15, p. 18).
Similar observations of cerebrospinal fluid circulation have been carried out in people, but with less invasive ways of measuring. In one, researchers from Oslo University Hospital, Rikshospitalet compared 15 patients who had a condition called normal pressure hydrocephalus, a kind of dementia caused by buildup of cerebrospinal fluid in the cavities of the brain, with eight people who didn’t have the condition.
The researchers used a tracer for cerebrospinal fluid and magnetic resonance imaging to measure the flow over 24 hours. Immediately after a night’s sleep, cerebrospinal fluid had drained in healthy people but lingered in the patients with dementia, the researchers reported in Brain in 2017.
Don’t snooze, you lose?
The central question — the one that doctors really want to answer — is whether better sleep could treat or even prevent Alzheimer’s. To try to figure this out, Bendlin and her Wisconsin colleagues are now studying people with sleep apnea. People with that condition stop breathing during the night, which wakes them up and makes for a lousy night’s sleep. A machine called a CPAP, short for continuous positive airway pressure, treats the condition.
“Once people start treatment, what might we see in the brain? Is there a beneficial effect of CPAP on markers of Alzheimer’s?” Bendlin wonders. “I think that’s a big question because the implications are so large.”
A study reported in Neurology in 2015 offers a reason to think CPAP might help. Using data from almost 2,500 people in the Alzheimer’s Disease Neuroimaging Initiative, researchers at the New York University School of Medicine found that people with sleep disorders like obstructive sleep apnea showed signs of mild cognitive problems and Alzheimer’s disease at younger ages than those who did not. But for those who used CPAP, onset of mild cognitive problems was delayed.
“If we find out that sleep problems contribute to brain amyloid — what that really says is there may be a window to intervene,” Bendlin says. And the solution — more attention to sleep — is one prescription with no side effects.
This article appears in the July 21, 2018 issue of Science News with the headline, “The Clean Cycle: The body may use sleep as a time to wash away the waste that can cause Alzheimer’s disease.”