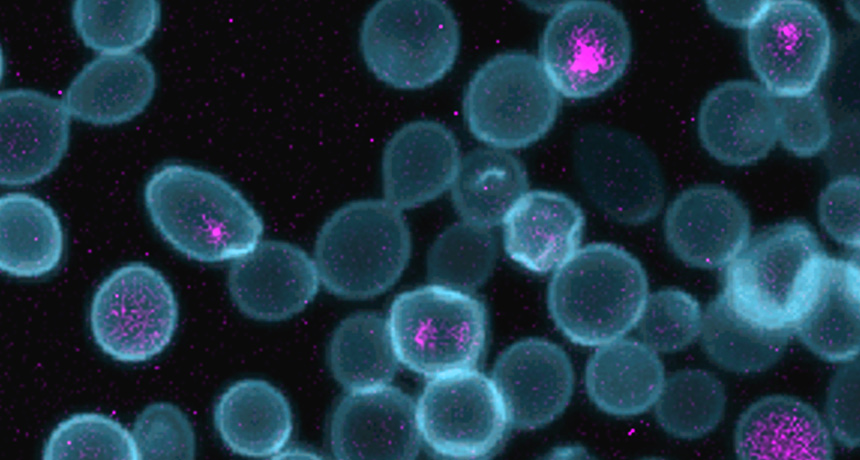
YOU COMPLETE ME Yeast cells (blue) harboring E. coli bacteria (purple) work together to provide each other energy and nutrients. The hybrid cells may help scientists learn more about the evolution of mitochondria, the organelles that make energy for cells.
Paul Webster