Diminishing Obesity’s Risks
Mouse data suggest that, properly managed, obesity can be benign.
Health-care professionals typically refer to an extremely heavy person as being morbidly obese. The term reinforces the idea that the individual is at high risk of diabetes, fatty-liver disease, and heart attacks. Researchers who have been working with mice now report that certain chronic diseases don’t have to be consequences of obesity.

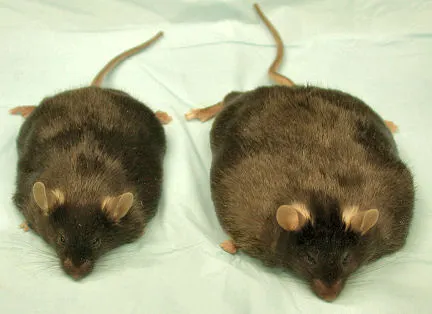
The team accomplished the disconnect by tricking the animals’ bodies into storing all their excess fat within their fat cells, or adipocytes.
That’s not what the bodies of rodents—or people—typically do. Initially, excess lipids—fat—are stored in these cells, making up what’s called adipose tissue or simply body fat. These deposits lie primarily in the breasts, belly, and thighs. However, once adipocytes fill up, new storage sites take up the overflow. Those new depots usually develop in muscle and the liver.
Of those two depots, the liver is more dangerous when it becomes fatty. Straightforwardly named, fatty liver disease can arise and lead eventually to hepatitis, cirrhosis, and death.
A drop in the hormone called adiponectin is the body’s signal to store fat outside adipose tissue. Sometimes referred to as the starvation hormone, adiponectin normally remains high in lean animals. With obesity, however, blood concentrations of the molecule fall.
Philipp E. Scherer of the University of Texas Southwestern Medical Center and his colleagues reasoned that keeping adiponectin concentrations high might fool the body into making extra adipocytes instead of sending surplus fat to muscles and the liver.
The team has now investigated the hypothesis in a strain of mice that make copious adiponectin regardless of how fat they become. In the Sept. 4 Journal of Clinical Investigation, the researchers report that as the novel mice mature, they become unbelievably huge. Indeed, muses Scherer, these are “the fattest mice ever reported,” with fat comprising 60 percent or more of their body weight.
As hoped for, the mice deposit all their excess fat in adipose tissue. Also in sharp contrast to other obese mice, the high-adiponectin animals develop no signs of diabetes. They also avoid a metabolic disorder known as syndrome X, which puts animals, including people, at high risk of heart disease (SN: 4/8/2000, p. 236).
So, although these barely mobile, blubbery mounds of flesh look like wrecks, they don’t appear to be at high risk for several chronic diseases associated with obesity, Scherer told Science News Online. Actually, he says, from the preliminary data, the mice “appear perfectly healthy.”
He suspects that there’s a lesson in this for investigators of human-obesity treatments. Drugs exist that raise adiponectin values in even overweight individuals. Most, like pioglitazone (Actos) and rosiglitazone (Avandia), are prescribed to treat diabetes. However, data suggest these drugs also reduce the buildup of fat in the liver.
Unfortunately, diminishing health risks in morbidly obese people may require far more than just supersizing their treatment with the diabetes drugs—especially since data reported earlier this year linked rosiglitazone with an increased risk of heart attack (SN: 6/23/07, p. 397).
Fat signals
Tissues throughout the body communicate on a regular basis via signaling hormones. Adiponectin is one of those messengers released by adipocytes to inform the rest of the body about how full the fat cells are. If they aren’t full, Scherer explains, the cells pour out copious adiponectin. The body then responds by directing its fat into those cells for storage. As adipocytes fill with lipids, they turn down the adiponectin signal, telling the body that it’s time to find new fat depots.
Adipocytes release several other messengers, among them leptin. As lipids swell the adipocytes, the cells crank up production of this hormone. Once released into the bloodstream, leptin circulates to the brain, where it offers a status report on how full the fat cells are. If leptin signals that there’s plenty of fat on hand, a healthy body not only experiences satiety but also reduces its food intake and burns more calories.
At some point, a spontaneous mutation in mice led to a strain of animals that lacked the ability to make leptin. The resulting rodents, always hungry and primed to store—not burn—any excess energy consumed, inevitably become obese. Scherer’s group worked with this strain and engineered it also to make extra adiponectin. The new mice typically produce about twice as much adiponectin as a normal, svelte rodent does. This excess is comparable to what can occur when people take certain diabetes-controlling drugs.
In the new study, the researchers compared normal, lean, leptin-producing mice with leptinfree, obese ones and the new leptinfree-but-high-adiponectin animals. By adulthood, the new mice far surpassed the girth of the original obese line. But instead of having high blood sugar and insulin concentrations—characteristics of the original obese animals that mimic type-2 diabetes symptoms—the new megafatties exhibited normal insulin and blood-sugar values. In fact, Scherer says, the engineered animals had about the same insulin characteristics as healthy, lean mice.
“That was a real surprise,” he concedes—”that the [new] mice could get so fat and yet remain very healthy, metabolically speaking.”
One solution: More fat cells
Most people are like obese mice, chronically taking in more calories than they burn, Scherer says.
Lipid buildup in the liver is “really the driving force for insulin resistance,” a metabolic change that precedes the development of diabetes, notes Scherer When this develops, the body makes normal amounts of insulin, but finds itself increasingly unable to use it. The end result: Too little insulin is used to move energy into cells, leaving high concentrations of sugar in the blood.
The new study with high-adiponectin mice shows that “if you can overcome this block of overexpansion of adipose tissue, there is no need for excess calories to deposit as fat in the liver,” Scherer says. Instead, fat can accumulate where it does the least damage, “in the professional fat-storage cell, the adipocyte.”
But Scherer doesn’t want to say that excess calories are benign when they wind up in fat cells. Bulging adipocytes send out a number of inflammatory compounds (SN: 2/28/04, p. 139). It’s not yet clear how important a role these compounds may play in chronic disease, but some have been linked to diabetes. Moreover, extra weight may strain an animal’s joints and even its heart. So, it’s premature to give a clean bill of health to mice whose physiques rival that of Jabba the Hutt.
Still, Scherer argues, “from a qualitative point of view, these [new] mice are relatively healthy.” Indeed, he says, what happens in the animals’ tissues may explain why some very obese people are able to retain good insulin sensitivity and dodge the diabetes bullet.
People who develop diabetes as adults tend to put all of their fat into a few big, inflammation-prone fat cells. However, some people’s bodies employ a different strategy, Scherer says. They pack relatively small quantities of fat into an ever-proliferating number of fat cells, ones that never seem to undergo stress-induced inflammation. This approach is triggered by a “local overexpression of adiponectin in adipocytes.” That, in turn, switches on production of a key signaling molecule—PPAR-gamma—that serves as a master switch “governing how many fat cells we have,” he explains.
“None of this is an endorsement for obesity,” Scherer cautions. “But it shows that if you can expand your fat stores in a healthy way to keep up with your caloric intake, this will improve insulin sensitivity.”
Overall, he argues, “the best strategy is to eat less and exercise more. But for the many of us who continue to take in more calories than we burn, it would be better to expand our fat-cell numbers than to store excess lipids in other tissues. That’s our take-home message.”
If you would like to comment on this Food for Thought, please see the blog version.







