How 4 major coronavirus tools impacted the pandemic in 2022
COVID-19 vaccines for the littlest kids and an updated booster marked Year Three of the pandemic
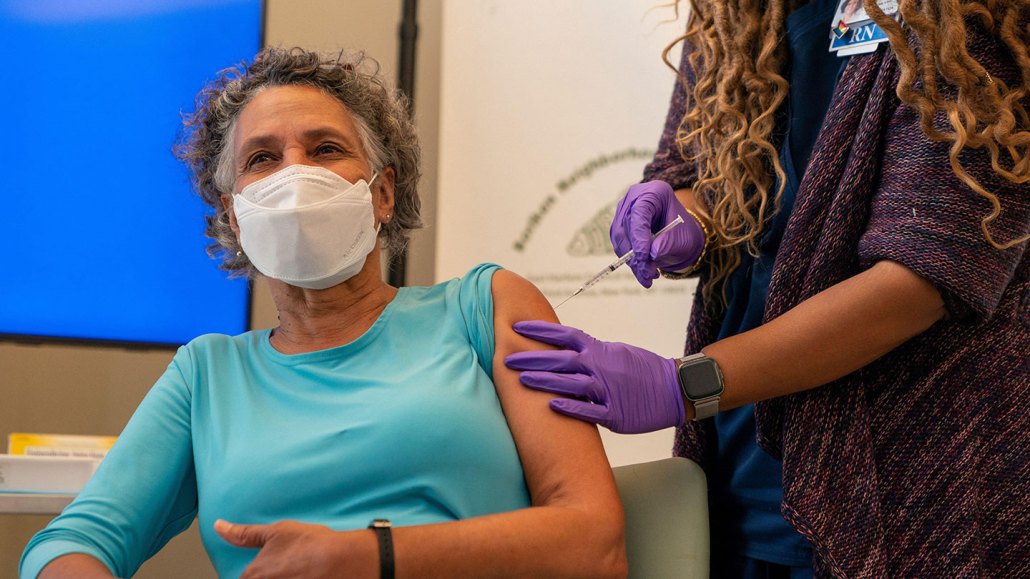
Mary Bassett, New York state’s Commissioner of Health, receives her updated booster in early September, soon after the shot was recommended. The booster was designed to spur an immune response against more versions of the virus.
Enrique Shore/Alamy Stock Photo