Alzheimer’s disease may come in distinct forms
Mouse experiments, if confirmed in people, imply that treatment should be personalized
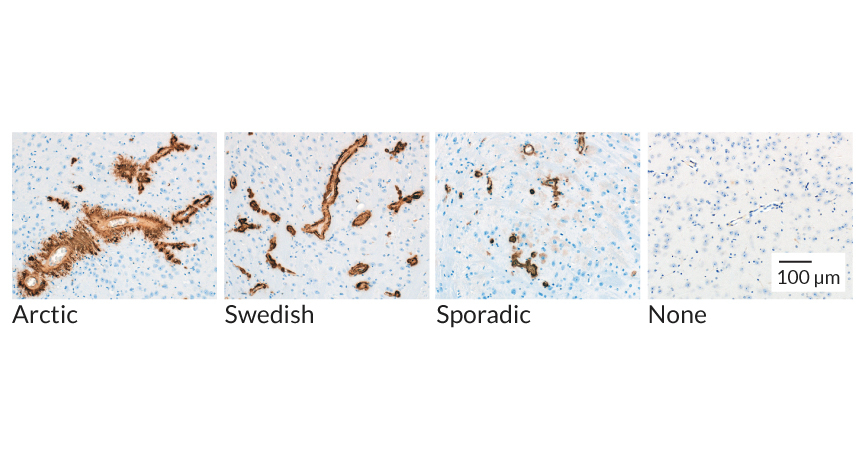
DISEASE DIFFERENCES Clumps of A-beta (brown) take on distinct shapes in the brains of mice injected with brain samples from people whose Alzheimer’s disease was caused by different mutations, called Arctic and Swedish, and by a sporadic form.
J.C. Watts ET AL/PNAS 2014
Alzheimer’s may come in multiple forms, two studies suggest. Distinct versions of a protein snippet called amyloid-beta reliably produce different effects in the brains of mice, scientists report June 30 in the Proceedings of the National Academy of Sciences.
The results are preliminary. But if similarly divergent pathways are found to be important in Alzheimer’s disease in people, the finding might ultimately enable custom-designed treatments, says neuroscientist Kurt Giles of the University of California, San Francisco, who was a coauthor of both studies. Currently, no treatment can halt or reverse Alzheimer’s-related brain damage.
Other researchers say that caution is warranted in interpreting the results. The lab studies on mice create “an artificial situation” that might not be relevant to human disease, says neurologist and neuropathologist Thomas Wisniewski of New York University’s medical school.
In one of the new studies, the researchers injected brain tissue taken from four people who had died with Alzheimer’s into mice engineered to produce the human form of A-beta. Two samples came from people with extremely rare mutations that caused their disease: one mutation called Arctic and one called Swedish. The other two samples were from patients with “sporadic” Alzheimer’s that, like most cases, arose with no known cause.
Once in the mouse brains, the Arctic and Swedish samples seemed to influence A-beta in different ways. Compared with the Swedish sample, the Arctic sample led to brains that had clumps of A-beta made of 38 amino acids, as opposed to clumps full of the more common forms with 40 or 42 amino acids. And Arctic A-beta fibrils appeared furry under the microscope and radiated outward from blood vessels in the brain. In contrast, Swedish fibrils appeared more compact and formed a single layer around the vessel. The sample from one of the people with a sporadic case of Alzheimer’s created effects in mice that appeared more similar to the effects of the Swedish sample, and mice injected with the sample from the other sporadic case displayed traits from both Swedish and Arctic samples.
These properties persisted even when the human samples were taken out of the mice’s brains and put into new mice. Scientists had previously seen some A-beta differences in a small number of people with the Arctic and Swedish mutations, but those differences may have reflected natural variation, Giles says. Showing that these traits persist from human brain to mouse to mouse is important evidence that these mutations represent strain differences.
In the second study, the researchers found that, like the samples taken from the four Alzheimer’s patients, distinct forms of A-beta created in the lab behaved differently once researchers set them loose in the brains of mice.
The two studies strengthen the much-debated claim that Alzheimer’s disease, Parkinson’s disease and other brain disorders stem from infectious misshapen proteins that incite nearby normal proteins to go rogue (SN: 12/29/12, p.24). These misshapen proteins are prions, Giles and his colleagues argue, a class of bad seeds famous for causing the brain-wasting Creutzfeldt-Jakob and mad cow diseases. Finding that different forms of A-beta can be transmitted is a hallmark of a prion disease, Giles says.
A-beta comes in several forms, he says, and people with Alzheimer’s probably carry complex mixtures of them. Many more human brain samples are needed to figure out whether and how these different forms of A-beta contribute to the disease. However, he says, “some of these brains, particularly with these mutations, are extremely difficult to get ahold of. We literally have to scour the world to get them.”
The results could ultimately lead to fine-grained Alzheimer’s diagnoses, depending on which sort of A-beta a person has, says coauthor of the studies Jan Stöhr, also of UCSF. And those fine-grain distinctions might allow targeted drugs, he says.
But those ideas will require much more research before they’re accepted. For now, there’s no evidence that Alzheimer’s disease can be influenced by different strains of A-beta, says neuropathologist Rudolph Castellani of the University of Maryland School of Medicine in Baltimore. Nor is there any evidence that A-beta behaves like a prion in people, he says. “The jury is still out.”