Arsenic Pollution Disrupts Hormones
Chronic exposure to small amounts of arsenic in drinking water increases a person’s risk of cancer and other diseases. Researchers believe they’re now onto an explanation for the process.
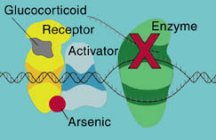
Arsenic acts as an endocrine disrupter, upsetting the action of hormones by blocking or mimicking them, say Joshua W. Hamilton of Dartmouth College in Hanover, N.H., and his colleagues. Previously identified endocrine disrupters have included many pesticides, industrial chemicals, and pollutants including dioxin. This is the first time that a metal has joined the list, says Hamilton.
The researchers suggest that arsenic interferes with the action of glucocorticoids, hormones never known before to be vulnerable to endocrine disruption by pollution. Glucocorticoids belong to the same family of steroid hormones as estrogen and testosterone. Glucocorticoids are responsible for turning on many genes that may suppress cancer and regulate blood sugar. Other researchers have identified estrogens, thyroid hormones, androgens, and melatonin as types of hormones that pollutants can affect (SN: 1/6/01, p. 8).
To test arsenic’s effect on the glucocorticoid system, Hamilton and his coworkers treated rat tumor cells with a solution containing water and a synthetic glucocorticoid, arsenic and the glucocorticoid, or water alone. They then measured the activity of a gene normally switched on by glucocorticoids. The hormone alone activated the gene, but the glucocorticoid-arsenic mix caused no more gene activity than the water solution did. Arsenic blocked the hormone’s action, the researchers report in the March Environmental Health Perspectives.
Ordinarily, hormones bind to a receptor in a cell, and the hormone-receptor complex then turns on genes. While other endocrine disrupters interfere with the binding process, arsenic alters the complex so it can’t turn on genes, says Hamilton. This may be the way some other endocrine disrupters work as well, he speculates. The new finding may help explain how arsenic triggers cancer, diabetes, and other chronic diseases such as hypertension, Hamilton adds.
Although arsenic at high doses kills cells immediately, this study shows that arsenic can be harmful even in low concentrations, says Alan R. Parrish of Texas A&M University in College Station. Hamilton’s team observed hormone-disrupting effects using arsenic concentrations comparable to 10 parts per billion (ppb) in water. “These are concentrations that people might be getting,” says Parrish.
Large geologic deposits of arsenic exist in Asia, South America, and several U.S. states, as well.
In January, the U.S. Environmental Protection Agency lowered the limit of arsenic in drinking water from 50 ppb to 10 ppb. “There’s a growing appreciation that arsenic has health effects at much lower doses than we originally thought,” says Hamilton.
The new finding suggests that arsenic affects hormones, but the work needs to be confirmed, says Stephen H. Safe of Texas A&M University. The researchers must show that arsenic acts similarly in other types of cells and in whole animals. “It’s not the end of the story, but it’s a nice lead,” says Safe.