Beat Generation: Genetically modified stem cells repair heart
In experiments on guinea pigs, scientists have used genetically modified human embryonic stem cells to make a biological pacemaker. The implanted tissue has kept the guinea pig hearts beating after their natural pacemaker cells were destroyed.
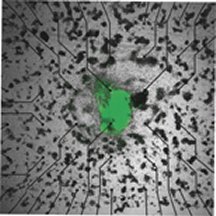
The researchers, led by Ronald A. Li of Johns Hopkins University in Baltimore, began with unspecialized embryonic stem cells. The scientists genetically modified them to produce green fluorescent protein. When they separated the cells into small clusters, the cells began to differentiate. Some clusters started to beat, indicating the presence of heart-muscle cells and pacemaker cells. The researchers then extracted cells from the beating clusters.
After transferring these cells into the animals, the scientists used a freezing probe to kill the animals’ own pacemaker tissue. Despite the loss of their natural pacemakers, the guinea pigs’ hearts continued to beat. Close examination showed that the rhythm was originating from the areas where the human cells had been implanted. Unlike battery-powered pacemakers, says Li, stem cell–based pacemakers speed up or slow down in response to drugs that alter normal heart rate.
The researchers aren’t sure why the guinea pigs’ immune systems didn’t destroy the intruding human cells. According to Li, the guinea pigs’ circulatory systems may not have thoroughly branched into the human cells and so the animals’ immune systems may not have detected them.
Li’s study, which appears in the Jan. 4 Circulation, isn’t the first to use stem cells to repair broken hearts, but it’s the first to do it with genetically modified cells. The resulting green glow made the human cells easy to spot in the guinea pigs, but more important, says Li, it’s proof that genetic modification didn’t disrupt the cells’ development.
“Now, we can move on to something more ambitious,” he says. His group has already learned how to genetically alter human pacemaker cells to fine-tune their firing rates, though the scientists have yet to test these modified cells in animals. Adjusting the firing rate could be important because ordinary pacemaker cells beat slowly when implanted. The heart’s natural pacemaker is a complex mixture of several cell types, Li says, so it’s difficult to mimic its function with any single, unmodified cell type.
Gordana Vunjak-Novakovic of the Massachusetts Institute of Technology, who investigates techniques for growing replacement heart tissue, says that although many groups are working to engineer heart tissue, Li’s study stands out because it investigates what happens after the engineered tissue is implanted. Still, she cautions that much work remains before heart-repair treatments can be developed for people. “We cannot even complete a list of hurdles at this time or predict how long it will take to resolve some of the problems we are working on,” she says.







