Breaking the Barrier
A technique combining ultrasound pulses with microbubbles may help scientists move therapeutic drugs across the brain’s protective divide
Normal 0 false false false MicrosoftInternetExplorer4
A blog entry on the Sussex Amateur Brain Surgery Club’s website boasts that “these days, brain surgery is very much the preserve of professional surgeons, but we at the Amateur Brain Surgery Club believe that anyone can do it, with a few basic tools and a little care.”
The post is a spoof, but whoever penned those words may be on to something.
The right tools could make treating the brain a simpler, less invasive process. Researchers have zeroed in on two such tools—sound waves and microscopic bubbles—that may eventually allow doctors to tackle a range of brain ailments, all without opening the head.
Although many medicines could work wonders for certain brain disorders, the majority of drugs can’t get from the blood to the brain. Tumor-shrinking compounds used in other parts of the body are too big to float through the tiny gaps in the brain’s blood vessels. So doctors treating brain tumors must still poke a hole in the head and cut out the offending mass. Without effective methods for drug delivery, promising treatments for brain ailments like Alzheimer’s have also stalled.
“People have calculated that about 97 percent of large molecules that could be useful in treating brain disorders cannot be used because they cannot get through the blood-brain barrier,” says Kullervo Hynynen, a medical biophysicist at the University of Toronto in Canada.
But researchers have been making inroads, finding ways to slip larger molecules into the brain’s tissue. Those deploying microscopic bubbles and ultrasound—sound waves above the audible range—to squeeze drugs through vessel walls and into the brain are leading this trend. The method has been tested only in animals so far. But if perfected, the technique could combat diseases like Alzheimer’s, Parkinson’s and cancer.
Keep out
In the late 19th century, bacteriologist Paul Ehrlich injected dyes into rats’ bloodstreams, hoping to gain insight into the structure of cells. But when he dissected the animals, he found a peculiar thing: While the liver, lungs and other organs were stained bright red, the brain didn’t soak up any color. He didn’t realize it then, but Ehrlich had inadvertently discovered the blood-brain barrier.
The barrier, a tightly packed layer of cells lining the brain’s blood vessels, protects the brain by keeping most molecules out. A molecular bouncer, the barrier uses special gatekeeping receptor molecules to actively escort in a select few chemicals, like energy-packed glucose, while denying entry to most large and highly charged molecules. The restrictive junction makes sure blood, toxins or nasty bacteria can’t enter the brain and wreak havoc. But when a person develops a tumor or Alzheimer’s disease, this same neural lockdown prevents doctors from getting medicine where it’s needed most.
More than a hundred years since Ehrlich’s work, methods for safely relaxing the barrier are still in short supply. Some chemicals, such as mannitol, can do the trick, but they have broad effects, opening the barrier across the entire brain, says neurologist Brian Bacskai of HarvardMedicalSchool and Massachusetts GeneralHospital’s campus in Charlestown. Mannitol, used to lower blood pressure inside the head, can also alter cellular function in other parts of the body. A few small molecules, such as those used to treat depression and epilepsy, can cross the divide. For the most part, though, getting medicine into the brain still requires opening the head and injecting drugs directly into the tissue.
Blowing bubbles
Since the 1950s, scientists have known that pulsing ultrasound through the brain can temporarily open the blood-brain barrier in animals, but only at intensities that cooked the tissue, says Hynynen, a pioneer in the field. For many years, every attempt to relax the barrier with ultrasound caused permanent brain damage.
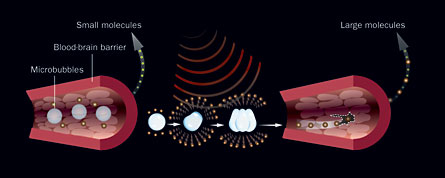
Then researchers looked to an existing ultrasound method—enhanced Doppler blood flow imaging. Like the sonar used by bats and ships, basic ultrasound images the inside of the body by measuring reflected sound waves. Doppler ultrasound shows movement, such as blood flowing through blood vessels. Clinicians can increase the sensitivity of the technique by injecting commercially available microbubbles into a patient’s circulatory system. Short ultrasonic pulses trigger the microbubbles to oscillate, enhancing the images.
Microbubbles can encase a gas or fluid within shells made of water-soluble proteins or fatty lipids. The secret weapon is “just like a soap bubble, only it’s stable in the bloodstream,” says Ralph Shohet of the University of Hawaii at Manoa, who uses ultrasound and microbubbles to deliver gene therapy to the heart, brain and pancreas.
The bubbles, typically a bit smaller than red blood cells, can travel through the circulatory system without forming an embolism—an air bubble that lodges in the heart, lungs or brain, Shohet says. What’s more, the FDA has already cleared microbubbles for clinical use.
By tweaking the existing Doppler approach, Hynynen and his colleagues have nudged the blood-brain barrier open. Their technique involves injecting microbubbles as well as a target drug into the blood vessels of mice or rabbits. The bubbles and drug quickly reach capillaries in the brain. The team then aims ultrasonic pulses at the microbubbles floating in the brain’s blood vessels, usually for several milliseconds.
Lengthening the burst duration from microseconds to milliseconds makes the blood-brain barrier permeable to the medicine.
“The difference between therapy and imaging is the pulse length,” says biomedical engineer Elisa Konofagou of ColumbiaUniversity.
In 2001, Hynynen’s group reported in Radiology that the microbubble-ultrasound combination could send molecules through the barrier without lasting damage. Focused ultrasound can also target regions just a few millimeters wide.
In animal models, the treatment made vessel walls leaky for up to six hours, Hynynen’s team found. After that, at least in those animals treated with the lowest power ultrasound, the barrier sealed up. Studying these animals’ brains under a microscope, researchers found no obvious signs of damage—no errant blood cells in brain tissue and no quirks in the shape or structure of nerve cells. At certain powers and frequencies, ultrasound and microbubbles seem to open up the brain in a reversible way.
Under pressure
Researchers haven’t pinpointed exactly how the technique causes reversible opening of the blood-brain barrier. The short ultrasound bursts don’t generate much heat, so people doubt the effect is thermal. Instead, mechanical forces may play a crucial role, says Kathryn Nightingale, a biomedical engineer at Duke University in Durham, N.C., who studies advanced ultrasonic imaging techniques.
Sound waves are simply localized waves of pressure variation. So when researchers target microbubbles with ultrasound, the bubbles shrink and expand in response to these changes in pressure.
When the microbubbles undergo these quick oscillations, the blood flowing around the bubbles gets squeezed and expanded too. The streaming blood appears to shear the nearby blood vessel wall, according to a paper in the June Advanced Drug Delivery Reviews. Hynynen’s team speculates that the forces caused by these streams may lead the receptor molecules in the barrier membrane to actively transport more molecules across the blood-brain divide.
The rapid pressure changes can also pitch the throbbing bubbles themselves against the vessel walls. This may stretch the tightly joined barrier cells, widening the narrow spaces between them and letting larger molecules sneak through.
At higher pressures, ultrasound forces bubbles to pop violently, creating shock waves that send jets of fluid slamming into vessel walls. The jets may poke tiny holes into the barrier that chemicals could pass through.
Many questions remain about how the microbubble-ultrasound duo opens the barrier and how to optimize the technique. Scientists are looking at, among other things, different sizes and motions of microbubbles—popping or vibrating—to better understand how to use the technique most safely and effectively.
Early research is beginning to offer clues. In past experiments, scientists had no way of knowing whether small or large bubbles were essential for opening the barrier. But Konofagou’s team has been sorting microbubbles by size and studying their effects separately.
Her group, reporting July 1 in Paris at the Acoustics ’08 meeting, found that bubbles larger than 4 micrometers—those that are more likely to expand and contract without popping—tend to produce larger open regions in the blood-brain barrier. When larger bubbles swell, they may get stuck in the capillaries and push on the vessel walls around them, Konofagou says. Smaller bubbles—which models predict are more likely to collapse violently—open smaller spaces.
And bubble collapse may do more harm than good. In the August Ultrasonics, Hynynen and his colleagues report that the blood-brain barrier can be opened without bubbles popping, which appears less damaging to nearby tissue. Popping bubbles have been linked in some studies to more red blood cells seeping into brain tissue, a sign of stroke.
Different diseases, same bubbles
A number of groups have harnessed this bubble behavior to test potential new therapies in animals. Shohet’s gene therapy research group combines ultrasound and microbubbles to ferry genes into brain tissue. Naked DNA injected into the bloodstream would get chewed up before it could enter a cell. But attaching a gene to the outside of a bubble protects the DNA until it reaches its target. So Shohet’s group tags the bubbles with DNA and sends them through the bloodstream into the brain’s vessels. Then the team hits the bubbles with ultrasound.
The collapsing bubbles pockmark surrounding cells. Part of a bubble’s shell juts through a tiny hole and hurtles a bit of the gene into the brain tissue, where the gene makes its way into brain cell nuclei, Shohet speculates.
Hynynen’s group uses focused ultrasound to deliver cancer-fighting compounds to the brain. One drug is Herceptin, an antibody used to treat breast cancer. Since breast cancer often spreads to the brain, the drug could be a powerful tool for battling secondary tumors. On its own, though, the bulky molecule cannot cross the barrier.
To get around this, the team injected microbubbles into mice and rabbits with brain tumors. Using MRI to locate the brain tumor, researchers directed bursts of ultrasound at the blood vessels near the tumor, opening the barrier locally and allowing the intravenously administered Herceptin to enter.
Both Bacskai’s and Konofagou’s groups use the new technique to study Alzheimer’s disease. Bacskai uses ultrasound to push larger chemicals into brain tissue for improved imaging. Konofagou’s team targets the hippocampus, a brain region important for memory. Her group hopes to move antibodies across the barrier that will target amyloid-beta deposits—clumps of toxic proteins implicated in Alzheimer’s. She also studies the substantia nigra region of the brain, where problems appear in Parkinson’s disease.
All these techniques require that the drugs—whether bound to microbubbles or not—be administered through an IV. This can cause problems with some chemotherapy medicines, which can enter various organs along the way to the brain, sickening the entire body.
Konofagou says that one approach to improve targeting would be to encapsulate therapeutics inside the bubbles. In this scenario, bubbles would sequester medicine, keeping it from acting on any other parts of the body. The drug could be activated only when the bubbles are hit with sound waves, so treatments could be delivered just to the brain tissue, where needed.
Obstacles ahead
Though promising, the method is “still a pipe dream,” Bacskai says. “But it’s not completely out of the realm of possibility.”
Researchers first need to show that the treatment is safe, Hynynen says.His group does this by carefully examining brain tissues from treated mice and rabbits. The team looks for signs of red blood cell leakage, any unhealed openings and damage to brain cells and the cells that make up the blood-brain barrier.
But such measures can’t reveal more subtle damage. Looking at the tissue of a dead animal is an imperfect measure of brain function, and tracking behaviors like grooming in mice can only uncover severe problems, Bacskai says. “Permeabilizing the membrane, even temporarily, is most likely going to have side effects you might not like, at least a headache,” Bacskai says. “But mice don’t complain about having a headache.”
Also unclear is whether getting drugs across the blood-brain barrier is enough. No one has shown that ultrasound-targeted drugs can penetrate nerve cells. Yet that’s where the drugs are most needed for diseases like Alzheimer’s and Parkinson’s, says Konofagou. “Just getting through the barrier is not the whole story.”
And though Hynynen can target cancer drugs to tumors, he hasn’t shown that treated animals live longer than their untreated counterparts. So his team has begun testing to see if the approach prolongs survival in mice.
Shohet believes scientists will also need to boost the amount of material reaching brain tissue. The tactic he uses to deliver gene therapy, for instance, “is a very inefficient method because only a little bit of the payload of the bubble is delivered,” he says. But a small amount of gene therapy can have big effects downstream.
Scaling the technique to human skulls may also stymie researchers, Shohet says. Ultrasound easily penetrates the delicate skulls of rats and mice. But thicker human skulls pose more of a challenge. Researchers may have to amp up the power of the ultrasound, which could increase the risk of tissue damage, he says.
Still, the need for barrier-breaking is all too apparent. Promising new drugs may be waiting in the wings, but most will be shelved until someone finds a safe way to get them across the divide.