Breathtaking Science
Biologists home in on the brain area that drives respiration
Nearly 2,400 years ago in a treatise aptly titled “On Breath,” Aristotle posed a question that continues to captivate scientists today: “How can we account for the maintenance of the breath inherent in us, and for its increase?” In a suburb just outside Washington, D.C., Jeffrey C. Smith shows just how close modern researchers are to answering that question. With the aid of a powerful microscope, a computer monitor, a loudspeaker, and an array of other devices, he and a colleague use a minuscule electrode to listen in on the electrical activity of a paper-thin disc of living brain tissue. Every few seconds, the speaker crackles with sound.
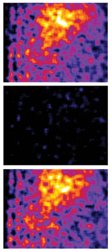
That recurring noise is compelling evidence that biologists have finally identified what French physiologist Jean Pierre Marie Flourens more than a century ago called the noeud vital—the vital node. It’s the source of a body’s natural breathing rhythm.
Over the past decade, Smith and other neuroscientists have homed in on this rhythm-generating kernel of nerve cells. They’ve located it within a well-defined region of the brainstem, the part of the brain that sits at the top of the spinal cord and controls most of the body’s automatic functions. This brainstem area, called the pre-Bötzinger complex, appears to drive both the normal, quiet breathing pattern and more complicated breathing actions such as sighs and gasps.
“We’re very far along the road of solving the basic problem of how we breathe,” says Smith, who works at the National Institute of Neurological Disorders and Stroke in Bethesda, Md. That solution is of more than academic interest. Understanding how the brain controls breathing could suggest new ways to prevent sudden infant death syndrome (SIDS), point toward safer anesthetics, and lead to better treatments for respiratory conditions such as sleep apnea, in which people have difficulty breathing while asleep. “There’s considerable clinical relevance,” says Smith.
The vital node
With two treatises on the topic, Aristotle was among the first to record his thoughts on the role of breathing. “A few of the earlier natural philosophers have dealt with respiration; some of them have offered no explanation why this phenomenon occurs in living creatures; other have discussed it without much insight, and with insufficient experience of the facts,” he dismissively noted. Aristotle, of course, had his own theory. He argued that breathing cools the body heat generated by the “fiery nature of the soul which exists in the heart.”
It was another 2,000 years before scientists began to recognize that breathing’s core function is to draw in oxygen from the air and expel carbon dioxide. Although people can hold their breaths for a while and sometimes exert great control over breathing, the process usually is on autopilot. And for good reason. “From birth until death, you can’t go too long without breathing,” Jack Feldman of the University of California, Los Angeles wryly noted in a lecture at the Society for Neuroscience meeting in Orlando last November.
A person’s heart can beat on its own, but it takes brainpower to drive even the automatic inhaling and exhaling of air. Galen, a Greek physician who lived a few generations after Aristotle, may have been the first to realize that. He noted that gladiators and animals could usually still breathe if injured below the neck but sometimes stopped breathing immediately when suffering neck and head injuries.
That observation points to the brain as the breathing center, but it doesn’t indicate a specific part of the brain. When investigators in the 18th century showed that rabbits continue to breathe after their cerebrum and cerebellum are removed, researchers began to concentrate on the brainstem. In the 1840s, Flourens narrowed the field’s focus to a specific brainstem region within an area called the medulla.
Lesions in this area, his noeud vital, stopped respiration in animals.
Over the next century or so, researchers tried to better define the boundaries of the noeud vital. Initially, they mostly performed lesion experiments, and then later they began recording the electrical activity of the brains of sedated cats.
“Breathing is the only movement that continues to be generated spontaneously by the brain in the deeply anesthetized animal,” notes Smith.
In the 1980s, researchers found that the neural basis of respiration could be studied in brainstems—attached to spinal cords—surgically removed from newborn rodents and kept alive in laboratory dishes. This tissue contains brainstem nerves that control muscles involved in breathing, such as the tongue and diaphragm muscles.
Even in a lab dish, these nerves continue to show periodic bursts of electrical activity that the scientists concluded reflect a basic respiratory rhythm. The bursts occur at a slower frequency than the normal breathing rate, says Smith, because the tissue is kept in oxygen-rich, lower-than-body-temperature conditions that reduce the typical bursting rate.
In the mid-1980s, Smith joined Feldman’s group and began an effort to further pinpoint the noeud vital. He painstakingly carved out pieces of the newborn rat brainstem and documented whether the respiratory rhythm persisted. In 1991, Smith, Feldman, and their colleagues reported that this rhythm could be traced to an area of the medulla near another brain region that controls breathing out. The region involved in exhaling had been named the Bötzinger complex after a German wine that scientists were drinking when they announced its discovery, so Smith and Feldman dubbed the newly recognized area the pre-Bötzinger complex (PBC).
The investigators also reported that nerve cells within this complex exhibit periodic bursts of electrical activity. Calling these cells “pacemaker neurons,” they proposed that a network of these cells is responsible for the rhythmic electrical activity observed in the nerves controlling respiratory muscles.
More recently, neuroscientists have sought a more molecular way to identify which nerve cells in the PBC generate the respiratory rhythm. In 1999, Feldman’s group reported rhythmic electrical activity in a set of about 600 PBC neurons that display a cell-surface protein that responds to a brain chemical called substance P.
Two years later, the scientists established the importance of those cells. They injected the PBC of live rats with a toxin that gradually destroys nerve cells bearing the substance P receptor. For a few days after the injections, the rats breathed normally. But after the toxin had killed more than 75 percent of the targeted nerve cells, the rodents began to breath so irregularly that they could not regulate the carbon dioxide and oxygen in their blood. Some of the rats died.
At the Society for Neuroscience meeting, Boston researchers investigating the origin of SIDS reported that the receptor for substance P may have led them to the human noeud vital. “We look at sites in the brainstem that are critical for the control of things like respiratory function, heart rate, and temperature control,” explains David Paterson of Children’s Hospital in Boston. “There’s a body of evidence suggesting that respiratory failure is part of the underlying cause of SIDS.”
The investigators studied the brainstems of five infants who had died of natural causes other than SIDS. Using an antibody marker that binds to the substance P receptor, the researchers found nerve cells that carry the receptor and are in a location in the infant brains roughly corresponding to that of the PBC in rodents.
“We still have a lot of work to prove that it’s the same region that we see in animals,” cautions Patterson. If it is, however, his team plans to investigate whether this brain region is abnormal in infants who died of SIDS or other respiratory disorders.
A sigh in a dish
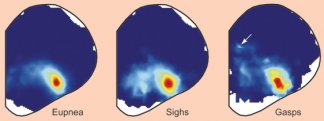
Does the PBC control all types of breathing? Yes, says Jan Marino Ramirez of the University of Chicago. He studies sighs and gasps, in addition to the typical, resting breathing pattern that scientists call eupnea.
A sigh is a breath with an extended period of inspiration, according to Ramirez. Most people think of a sigh as the loud exhaling that follows this intake of air, he notes. Infants tend to sigh about every 10 minutes, but this frequency goes down as a person grows up.
“The sigh is an arousal mechanism. Ninety percent of the time when you wake up at night, [it’s] due to a sigh or starts with a sigh,” notes Ramirez.
On the other hand, a gasp is a much more rapid, dramatic intake of air. The gasp, says Ramirez, is largely a last-ditch effort to bring in air when the body is starved for oxygen.
There’s evidence that infants who die of SIDS don’t sigh or gasp normally. “Basically, the two emergency mechanisms, the sigh and the gasp, are reduced [in SIDS babies],” says Ramirez.
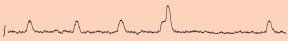
About 2 years ago, he and his colleagues reported that sighs and gasps can be discerned within the respiratory rhythm recorded with electrodes from rodent brainstem tissue. Whereas eupnea shows up as single bursts of electrical activity, occasionally a second, larger burst immediately follows this regular burst. Ramirez considers this a sigh. Moreover, if the rodent brainstem is subjected to low oxygen availability, the respiratory rhythm occasionally exhibits spasms of electrical activity that are briefer but much stronger than a typical burst. Ramirez interprets those powerful surges as the equivalent of gasps.
When the investigators looked to see where each of these different bursting patterns originated in the brainstem, they found that the electrical activity always centered within the PBC. These findings stunned Ramirez. He had long thought that sighs and gasps were triggered by networks of nerve cells distinct from the one that controls normal breathing. Now, he argues that a single neural network simply reconfigures itself to produce different kinds of respiration. “The same network can generate normal breathing, gasping and sighing,” he says.
Finding the rhythm
Not everyone is convinced by the current evidence pointing to the PBC as the neural locus of breathing. Some scientists still argue that a much larger network of nerve cells drives the respiratory rhythm. Even Smith and Feldman acknowledge that there’s much more to the neural control of breathing than the PBC.
“The evidence is pretty unequivocal that this is a key element for the generation of respiratory rhythm. But in an intact animal, there are other things going on” that influence this rhythm, Feldman says. “You might need an engine in a car, but you can’t drive down the street with just the engine.”
Donald McCrimmon of Northwestern University in Evanston, Ill., says that few scientists question that the rhythmic activity of the PBC in a laboratory dish reflects some aspect of breathing. “But how you really build a robust respiratory-control system—on top of this [complex] that generates a rhythm—is the really big question,” he adds.
An equally big question for McCrimmon and others who embrace the PBC as the noeud vital is how the basic respiratory rhythm of eupnea arises, seemingly from fewer than 1,000 nerve cells. More than a decade ago, when Feldman and Smith first proposed that the periodic electrical bursts seen in pacemaker neurons could drive that rhythm, that model made sense to many scientists. Recently, however, Feldman has developed reservations about it.
“It was such a compelling and straightforward explanation that it rose from hypothesis to paradigm before one had the chance to do the experiments needed to test it,” he says.
In the May 30, 2002 Neuron, Feldman’s group offered data reinforcing his doubts. The investigators reported that treating the PBC in rodent brainstems with a drug called riluzole didn’t alter the recorded respiratory rhythm. That surprised Feldman because riluzole blocks the periodic electrical bursts of pacemaker neurons by inhibiting cellular pores known as ion channels. He and his colleagues concluded that the pacemaker bursting isn’t at the heart of the respiratory rhythm.
However, that result hasn’t been confirmed. Smith’s group recently conducted similar experiments and found that riluzole treatments do indeed interrupt the respiratory rhythm recorded from rodent brain tissue. And Ramirez has evidence that riluzole can block the gasping rhythm, but not the normal breathing rhythm. His studies suggest that there may be different kinds of pacemaker neurons within the PBC.
These discrepancies may be related to the way the brain tissues are sliced and exposed to the drug, suggests Smith. “It’s certainly something that’s going to be resolved over the next few months,” McCrimmon says.
One node or two
It may take longer to address the tantalizing question of whether there’s a more primitive noeud vital within the mammalian brain, an issue Feldman broached at the Society for Neuroscience meeting.
He noted that opiates slow a person’s breathing rate, which can be a considerable problem for physicians administering opioid-based narcotics. In still unpublished work, Feldman’s group found that rats don’t respond to opiates by gradually breathing slower but instead by skipping breaths.
Feldman interprets his group’s data as evidence for two sources of respiratory rhythm within the brainstem, an opioid-insensitive one with a naturally slow breathing rate and the PBC, which is opioid-sensitive and generates a faster respiratory rhythm.
Japanese researchers have recently identified a brainstem region near the PBC that exhibits a respiratory rhythm and is insensitive to opioids, Feldman notes. “Our current thinking is that the PBC in mammals is the dominant rhythm generator, but one has to consider the role of this second group of cells,” he says.
Feldman speculates that this newfound area appeared early in vertebrate evolution to drive the slower respiratory rhythm of fish, amphibians, reptiles, and other animals that don’t have a diaphragm. But the faster-paced PBC arose in mammals as they evolved a bellows-like diaphragm and had a greater demand for oxygen, he suggests.
A second noeud vital would add a new dimension to studies of the neural control of breathing. Still, with the PBC in hand, researchers remain confident that they are close to explaining one of the most fundamental examples of how the brain generates a behavior.
“There’s a reason to hope that in the not too distant future, we’ll be able to say that this is the basic [brain] circuitry for generating the respiratory rhythm,” says McCrimmon.
****************
If you have a comment on this article that you would like considered for publication in Science News, please send it to editors@sciencenews.org.