Cancer cells cast a sweet spell on the immune system
Researchers try to wake up immune cells by focusing on the sugars on the tumor surface
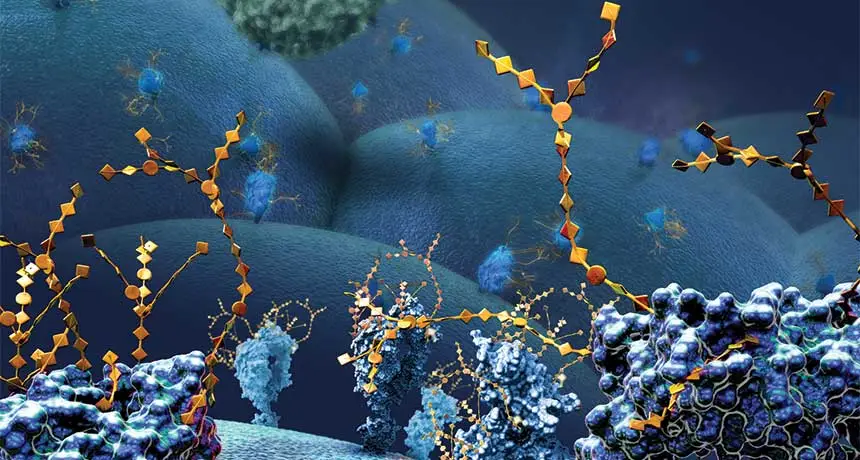
CANCER'S SWEET CLOAK Large surface proteins with chains of sugars (illustrated, yellow) on the outside of a cancer cell hide the tumor from immune attack and lull the immune system into a do-nothing trance.
NICOLLE RAGER FULLER
Shrink yourself small enough to swoop over the surface of a human cell, and you might be reminded of Earth’s terrain. Fats, or lipids, stay close to the surface, like grasses and shrubs. Proteins stand above the shrubs, as mighty oaks or palm trees. But before you could distinguish the low-lying lipids from the towering proteins, you’d see something else adorning these molecules — sugars.
If proteins are the trees, sugars are the mosses that dangle from the branches or, perhaps, the large fronds of the palm. “The cell surface is basically coated with sugars,” says Carolyn Bertozzi, a chemist at Stanford University. “They’re what viruses, bacteria and other cells see first when they touch down on a target cell.”
The sugars that attach to cell-surface proteins and lipids often take on elaborate structures. Information encoded within these structures helps cells recognize each other and relay messages in virtually every tissue and organ system. Yet compared with life’s other building blocks — proteins, lipids and nucleic acids — sugars languish in research obscurity. Most sugars are so complex that many scientists are ill-equipped to understand the basic biology, Bertozzi says. “They literally steer their research program in a different direction.”
Not Bertozzi. She’s among a small crew of scientists who have spent their careers steeped in sugars. Some of their studies have led to a new insight into cancer therapy: Manipulating sugars on the surface of tumor cells has the potential to expand an exciting new class of cancer drugs.
Much of the current optimism about fighting cancer focuses on immune therapies — drugs that awaken the body’s defenses to attack tumors (SN: 12/27/14, p. 8). The therapies focus on blocking protein interactions that suppress the immune system (SN: 7/11/15, p. 14). Such treatments have helped thousands of patients with cancers once untreatable, including former President Jimmy Carter, who was treated in 2015 and 2016 for advanced melanoma.
Story continues below slideshow
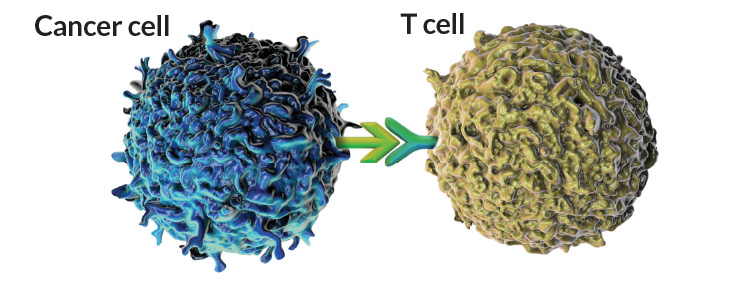
Surface tension
Cancer cells have surface proteins and sugars that link with immune cells to quiet their reaction to the cancer. Current immune therapies block interactions between PD-L1 proteins (light green) on tumor cells and PD-1 proteins (teal) on T cells to awaken the immune system to the tumor. A new wave of therapies could target parallel interactions between sialic acid sugars (orange) on the tumor and sugar-binding Siglec proteins (purple) on natural killer cells and other innate immune cells.
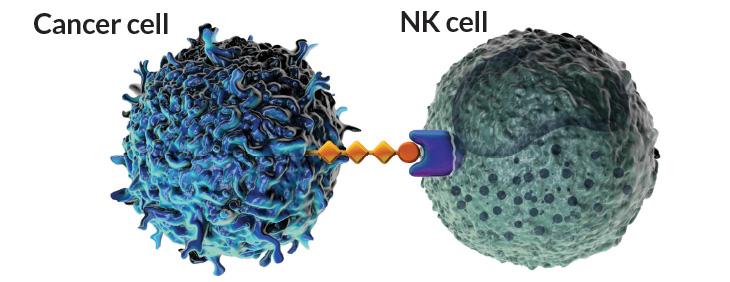
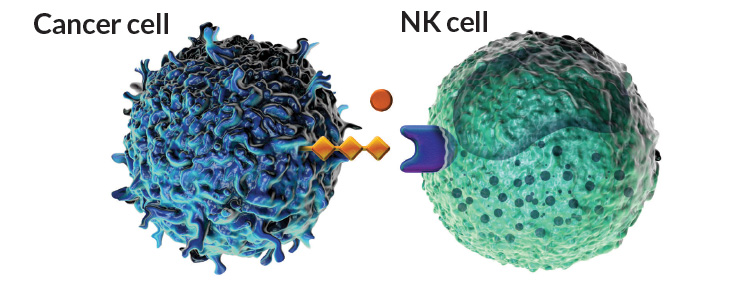
Source: V.H. Trang and P.D. Senter/PNAS 2016
But so far these drugs, called checkpoint blockers, work only against some cancers, such as melanoma, kidney cancer and non-small cell lung cancer, and not for all patients. An estimated 10 to 20 percent of cancer patients who get the drugs show improvement.
A new wave of potential immune therapies aims to target cell-surface sugars instead of proteins. Researchers hope these new approaches will rouse new warriors — an entirely different part of the immune system — to the fight. Current protein-targeting immune therapies activate killer T cells, part of the adaptive immune system with a memory for intruders and an ability to respond to specific threats.
Sugar-targeting drugs, on the other hand, would alert cells of the innate immune system, such as natural killer cells, or NK cells, and macrophages. Innate immune cells also help defend the body against bad guys, be they flu viruses or cells that have turned cancerous, but the innate cells kick into action more quickly and with less specialized responses than T cells. The two systems are complementary.
Clever disguise
Knowing what to attack requires immune cells to distinguish self from foreign, and sugars play a key role. One class of surface sugars, called sialic acids, mark the body’s cells as “self.” Sialic acids have a range of biological functions — their roles are so essential that preventing synthesis of these sugars in mice kills the animals in utero.
To our defense system, cell-surface sugars are a molecular fingerprint, telling a roving immune cell, “This one’s OK. Move along,” Bertozzi says. Certain pathogens, such as the bacteria that cause gonorrhea or streptococcal infections, have taken advantage. They coat themselves with sialic acids to hide from the immune system. Several years ago scientists wondered if tumors might use a similar trick. “We thought, ‘if bacteria are doing that, maybe cancer cells do too,’ ” says glycobiologist Ajit Varki of the University of California, San Diego.
That suspicion has roots in a strange but widespread observation researchers had made many years before: Sialic acids cluster in unusual ways on the surface of tumor cells. The observation intrigued Bertozzi. In the late 1990s, while starting up her lab at the University of California, Berkeley, she saw sialic acids as a potential marker for cancer. An expert in designing chemical reactions to tag molecules within living systems, Bertozzi thought about developing an early detection test that measured excess sialic acids.
Paper after paper came out linking cancer with an overgrowth of cell-surface sugars. However, none of the studies explained what triggers the ramp-up of sialic acids or why it matters. “What was the biological consequence? We didn’t know,” Bertozzi says. So her cancer detection idea sat on the back burner.
Then Bertozzi came across a 2010 paper in the Journal of Immunology that suggested there was more to the story. In that study, researchers in Israel used chemicals to cause tumors in mice bred to have weak immune systems. Tumors growing in these animals had lower levels of sialic acids, compared with tumors induced in normal mice. In separate experiments in lab dishes, the researchers stripped sialic acids off the surface of tumor cells and saw natural killer cells wake up and attack the cancer. To the researchers, the connection was stunning: Sialic acids seemed to protect tumors from the immune system.
Connecting the dots
While Bertozzi puzzled over sialic acids, a few other research teams had already spent decades studying immune cells that recognize and bind to these sugars.
Paul Crocker, an immunologist at the University of Dundee in Scotland, studies macrophages, part of the early responding, innate immune system. Greek for “big eaters,” macrophages feel their way with sticky spaghetti-like arms to find and devour pathogens and dying cells. In 1986, Crocker’s team discovered a protein that makes macrophages sticky. The researchers later named it sialoadhesin because it binds to sialic acids on the surface of other cells. Checking for signature DNA sequences in the gene that codes for sialoadhesin, researchers were excited to discover the protein wasn’t a lone ranger — it’s part of a large family of proteins already known to help immune cells signal to each other. “That was a breakthrough moment,” Crocker says.
In 1993, Varki and colleagues found a different set of immune cells, from the adaptive immune system, that also recognize sialic acids. B cells, which help call T cells into action, have a surface protein called CD22 that binds to cell-surface sialic acids.
A flurry of studies turned up more immune cell surface proteins with similar features. A total of 14 proteins, known as “Siglecs,” now make up this group of immune cell molecules that bind to sialic acid sugars. The founding members, sialoadhesin and CD22, are known as Siglec-1 and Siglec-2.
When Siglec-7 was discovered on natural killer cells, Crocker’s attention went to cancer. Guarding against tumors is “what NK cells are famous for,” he says.
By 2014, a trio of papers confirmed that Crocker’s shift to focusing on cancer made sense. Varki’s lab and a team led by Stephan von Gunten of the University of Bern in Switzerland analyzed biopsy material from people with various cancers. These studies showed that sialic acids on patient tumors indeed bind to Siglecs on human innate immune cells.
Once again, the cell-surface sugars acted like a cloak. When sialic acids bind to Siglecs on the surface of NK cells, these immune cells lose their cancer-fighting ability. That fact was shown in a common cell culture test that mixed immune cells with radioactive tumor cells. When NK cells attacked, the tumor cells burst and released radioactivity. But when NK activity was weak, less radioactivity leeched out.
Bertozzi and colleagues provided further support for the cloaking idea by working out a method to adjust the level of sialic acids on cells. The system was artificial — synthetic sugar molecules planted on the surface of human cancer cell lines — but it clearly showed cause and effect, she says. By dialing up sialic acids, the researchers could protect cells from NK killing. The team shared its findings in 2014 in Nature Chemical Biology.
With evidence from multiple labs suggesting that tumors cloak themselves in sialic acids to thwart the immune system, it was time to think about devising treatments that awaken the immune cells from their sugar-induced stupor.
Unpredictable diversity
Sugars far outnumber proteins among surface molecules responsible for distinguishing tumor from normal, Varki says. So you’d think they would make easier drug targets. Trouble is, sugars are immensely harder to study than proteins.
Both proteins and sugars adopt three-dimensional shapes to do their work. Once researchers have a protein’s genetic blueprints in hand, they can predict its structure. Sugars, on the other hand, have no clear templates. A sugar’s shape and structure are determined by a sequence of enzymatic reactions that can be hard to predict. Different cells turn on different sugar-modifying enzymes, so the same sugar can adopt a range of conformations. The “degree of complexity is thousands, if not millions, times that of proteins,” Varki says.
These structural differences affect how well sugars bind to proteins and lipids. And those interactions can have life-or-death ramifications, such as shielding cancer cells from immune attack.
Sugars are the “dark matter” of the biological universe, Varki says. They “affect everything, but hardly anybody studies them.”
Another checkpoint
To further complicate matters, sugars such as sialic acid are just one part of a tumor’s cell-surface barcode. Other players such as proteins also tell immune cells whether to attack or move on. The good news is that current immune therapies awaken immune cells to fight cancer by disrupting some of these proteins. Bertozzi and others think they can apply similar strategies to target cell-surface sugars.
Protein-based immune therapies work by deactivating cellular “off” switches on immune cells. One off switch involves a protein called PD-1 on the surface of T cells. When PD-1 latches onto the protein PD-L1 on a tumor cell, the T cell is turned off, rendered powerless against the cancer.
Siglecs play a similar role to PD-1, but mostly on natural killer cells and macrophages rather than T cells. The sialic acids on tumor cells work like the PD-L1. Just as existing immunotherapy drugs block PD-1, it’s conceivable to design new therapies to block Siglecs so that innate immune cells won’t be turned off. Indeed, biotech companies are pursuing these and other sugar-centric approaches.
But blocking too many of the immune system’s off switches could be dangerous. They have an important job: guarding against overzealous T cell activity that could trigger autoimmune disease.
Rather than go after Siglecs on immune cells, Bertozzi and colleagues are focusing on the sialic acids on the tumor cells. As long as a tumor cell wears a thick coat of sialic acid, immune cells won’t touch it, she says. “If you could strip off those sugars, the immune system could see the cancer cell for what it really is — something that needs to be destroyed.”
Many organisms, including humans and some microbes, have an enzyme that clips sialic acids off the ends of larger carbohydrate, protein or lipid molecules. Some versions of these enzymes, called sialidases, are commercially available as lab reagents. Bertozzi’s group decided to make its own by purifying large batches of the enzyme from bacterial cultures.
The trick was finding a way to point the sialidase specifically at tumor cells without letting it cut willy-nilly everywhere else.
Cancer cell editing
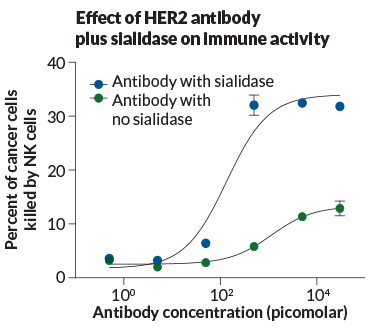
To get the sialidase to focus on tumor cells, Bertozzi and colleagues repurposed Herceptin, a blockbuster cancer drug. Herceptin is an antibody that recognizes a protein called HER2 on the surface of many breast tumors. When the antibody binds to HER2, it marks the tumor cell for destruction by innate immune cells, such as natural killer cells and macrophages. And since Herceptin binds to tumor cells better than normal cells, Bertozzi figured it could work as a delivery device to bring sialidase to tumors.
Using a method her lab had developed for joining antibodies to small molecules, Bertozzi’s team fused a sialidase enzyme with the Herceptin antibody. The researchers added the antibody-enzyme combo into a mixture of breast cancer cells with varying levels of HER2 on the surface. When the drug latched onto HER2 proteins on the tumor cells, the sialidase trimmed sialic acids like a chemical lawn mower, Bertozzi explains.
The strategy — published in September in Proceedings of the National Academy of Sciences — looks promising in these lab dish experiments, but it will take additional refining before the approach can be tested in people. One safety consideration relates to the antibody’s target, HER2. Though it is found in excess on breast cancer cells, the protein is also found at low levels on normal cells. So it’s possible the “lawn mower” could stray and trim sialic acids from cells that need the sugars for proper function.
Another concern: The engineered molecules contain bacterial sialidase, which — like other foreign proteins entering our bodies — would probably trigger a vigorous immune response. The antibody-enzyme combination “is a good proof of principle” showing it’s feasible to edit molecules on cancer cells, Varki says. “But I don’t think [the injections] would be practical to do repeatedly.”
Bertozzi is thinking along similar lines. Since publishing version 1.0 of the chemical lawn mower, her lab group has created version 2.0 and is now testing it in animal models. Version 3.0 will fuse antibodies with human sialidase, which is less likely to trigger an immune response, Bertozzi says. Her team is also working to attach sialidase to antibodies attracted to molecules other than HER2, to direct the enzyme to different kinds of tumors.
Two-faced macrophages
Meanwhile, other secrets lurk within the sugar coating on tumor cells. One of them emerged in a study published in November in Nature Immunology. A team led by biologist Joy Burchell of King’s College London was studying a large protein studded with sugars, including sialic acids. Called MUC1, this protein adorns the surface of normal epithelial cells found in the lungs, stomach, intestines, eyes and other organs. Modified forms of MUC1 appear on various cancer cells. Siglec-9 on the surface of macrophages is known to bind to tumor-specific versions of MUC1.
When those odd MUC1s bind to Siglec-9 on macrophages, the immune cells are thrown into a trance, the researchers found. The binding quiets macrophages in such a way that they ignore the tumor cell. Researchers had known macrophages adopt this weird state around tumors, but this study shows that MUC1 drives the transformation.
Not only does the MUC1–Siglec-9 interaction seem to shut down macrophage activity, it makes macrophages, which have some PD-L1, display even more. The finding raises questions because PD-L1 is a target of some immune therapies.
Current immune therapies unleash T cells to attack tumors by blocking the off-switch interactions between PD-1 and PD-L1. Focusing on the innate immune system could also help, and this paper suggests a possible target, Burchell says. She plans to work with a company to test, among other things, if blocking Siglec-9 can slow tumor growth in mice. And Varki cofounded Siamab Therapeutics in Newton, Mass., to design therapies that target other abnormal sugars on cancer cells. None of these drugs have entered human testing.
The under-the-radar work on sialic acids over the last few decades may have exposed just the edge of a dense forest. It’s likely that other sugars also form cloaks to shield tumor cells from immune attack, Bertozzi says. Translating such insights into actual therapies will require biologists to jump into the sugar fray and venture deeper than ever before. Bertozzi is hopeful: “We might be on the cusp of entering that phase.”