

Women may want to reconsider that popular style accessory, certain hard plastic water bottles available in fashion-coordinating colors. New animal studies link the chemical bisphenol A, which leaches from such polycarbonate plastics and food can linings, with heart arrhythmias in females and permanent damage to a gene important for reproduction. Other recent research suggests that human exposure to BPA is much higher than previously thought.
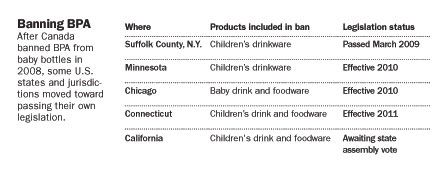
In animals, fetal exposures to BPA can be especially risky, sometimes fostering brain, behavioral or reproductive problems (SN: 9/29/07, p. 202). Canada and some states are moving to ban polycarbonate plastic in baby bottles for that reason. But the new heart data suggest that even adult exposures to BPA might cause harm.
In one new study, researchers treated mice with BPA during the middle of their pregnancies. All female offspring of the treated mice suffered an irreversible genetic change in one of the “master regulatory genes” of fertility, Hugh Taylor of the Yale School of Medicine reported in June in Washington, D.C., at the annual meeting of the Endocrine Society.
This gene, HOXA10, orchestrates the activity of “hundreds — if not thousands — of downstream genes,” Taylor says. Through the genes it controls, HOXA10 helps synchronize the timing of uterine changes and ovulation. Without that synchrony, “you won’t get pregnancies,” he explains.
The HOXA10 gene lost a methyl group (a carbon bound to three hydrogen atoms), permanently altering its activity and rendering uterine tissue hypersensitive to the effects of estrogen.
That’s probably not good, Taylor says, because “many diseases we see in adults owe their origins to fetal exposures” — when genes become inappropriately modified.
In another study presented at the endocrine meeting, Scott Belcher of the University of Cincinnati and his colleagues reported that BPA boosted “pro-arrhythmic activity” in isolated muscle cells from mice and rats.
Arrhythmias, or heartbeat irregularities, are blamed for a higher mortality rate after heart attacks in premenopausal women compared with men, Belcher says.
During pregnancy, vulnerability to heart arrhythmias rises with higher estrogen levels. Belcher’s team found that in these cells, BPA’s effect on arrhythmia risk was nearly identical to estrogen’s.
In whole rat hearts exposed to BPA or estrogen, pockets of cells refused to beat in concert with others, his group showed, setting up potentially life-threatening arrhythmic events. The problem escalated dramatically when female hearts were exposed to both estrogen and BPA.
Belcher’s group traced the effect to a certain type of estrogen receptor called the beta form, which is more active (and more abundant) in women. The scientists linked this receptor’s activity to a leading cause of arrhythmias — a leak of calcium from a part of heart cells known as the sarcoplasmic reticulum.
These data suggest that at estrogen levels typically found in premenopausal women, the addition of BPA would probably spike vulnerability to arrhythmias, Belcher says. In postmenopausal women, where estrogen levels are naturally low, it’s possible that BPA might also boost arrhythmia risk. “We’re doing studies in whole animals to address that,” Belcher says.
Although a broad host of animal studies have linked BPA to adverse health effects, comparable human data do not exist. Any human risks would depend on how much BPA actually gets into the body.
To probe one BPA source, Karin Michels of the Harvard School of Public Health recruited 77 undergraduates to consume all their cold drinks for a week out of stainless steel bottles. The next week, the participants drank from polycarbonate alternatives. Michels’ team sent students’ urine samples to a lab at the Centers for Disease Control and Prevention to assay BPA levels.
Even in the first week, when drinking from steel bottles, most students showed measurable levels of BPA. Those concentrations rose by 69 percent in the second, polycarbonate week to 2.0 micrograms per gram of creatinine, a waste product in urine, the researchers report online May 12 in Environmental Health Perspectives.
“I went in expecting that we’re so overwhelmed by BPA from other sources that this one variable would not make a difference,” Michels recalls. “But it did. That’s just amazing.”
A Canadian government study found another source of BPA — jars of baby food. They traced the BPA, present at levels of only parts per billion, to the resin that coats the underside of lids, Health Canada scientists report in the June 24 Journal of Agricultural and Food Chemistry. BPA-based resins line most food and beverage cans.
Studies have indicated that food is the dominant source for most people and that any BPA ingested from food should peak in blood within four hours, then quickly be excreted. However, there are growing suspicions that previous studies have underestimated how long BPA lingers in the body.
“By 12 to 18 hours [after eating] it should be practically gone,” says Richard Stahlhut of the University of Rochester Medical Center in New York. “For years that‘s been almost a mantra.”
But when his group looked at residues excreted by participants of a recent National Health and Nutrition Examination Survey — 1,469 adults who fasted for five to 20 hours before giving urine — there was still about as much BPA excreted 12 to 20 hours after a meal as just five hours after eating, the researchers reported in the May Environmental Health Perspectives.
This finding could mean there are major sources of BPA contamination other than food, Stahlhut says. More likely, he now suspects, a substantial amount of the BPA that enters the body may temporarily collect in fat, then slowly empty back into blood and become excreted. His team is now exploring this possible explanation.
The U.S. Food and Drug Administration estimates typical daily human BPA consumption at roughly 0.1 micrograms of BPA per kilogram of body weight. But when Frederick vom Saal of the University of Missouri-Columbia and colleagues administered 4,000 times that much to 11 rhesus monkeys, BPA blood residues in the spiked monkeys ended up only one-eighth as high as seen in a German study of pregnant women, he reported at the Endocrine Society meeting.
If these monkeys metabolize BPA at rates comparable to people, vom Saal says, then “humans would have to be exposed to over 1,000 micrograms per kilogram per day in order to achieve the kind of [blood] levels that are seen in multiple studies, not just the [German] one.” The FDA and the Environmental Protection Agency have estimated that a safe upper limit for daily human consumption of BPA is only 50 µg/kg of body weight per day.
Based on such data, House Committee on Energy and Commerce chairman Henry Waxman (D-Calif.) and the committee’s Oversight and Investigations Subcommittee chairman Bart Stupak (D-Mich.) sent a letter on June 2 to FDA Commissioner Margaret Hamburg. It asks FDA to “reconsider the Bush administration’s position that BPA is safe at current estimated exposure levels.”






