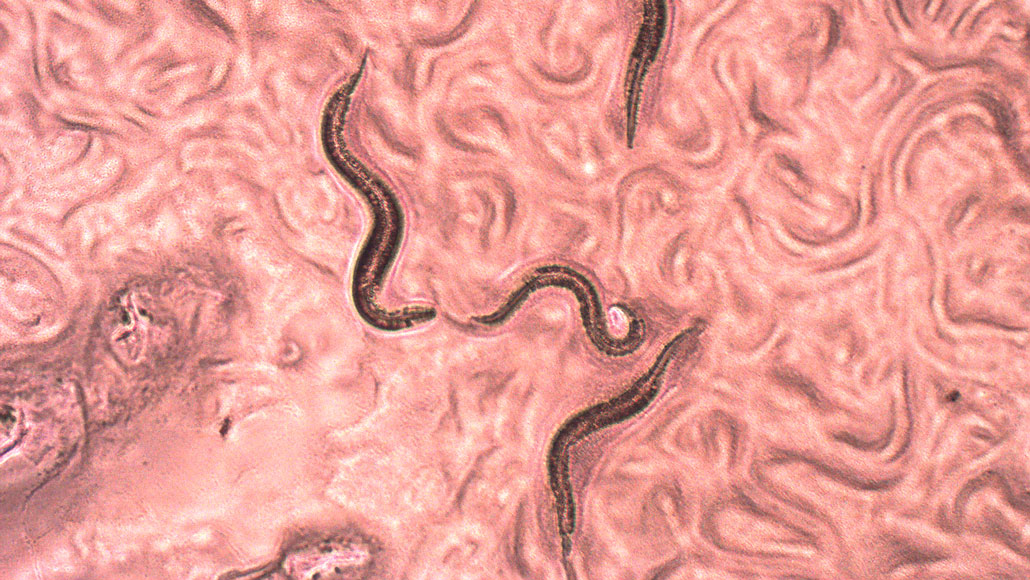
Named after Mephistopheles, a devil in Faustian legend, devil worms (Halicephalobus mephisto) live in hellish conditions up to 1.3 kilometers underground.
John Bracht and Megan Guerin

Named after Mephistopheles, a devil in Faustian legend, devil worms (Halicephalobus mephisto) live in hellish conditions up to 1.3 kilometers underground.
John Bracht and Megan Guerin