A Different Side of Estrogen
Second receptor complicates efforts to understand hormone
The mice in Jan-Åke Gustafsson’s lab are obese, their bones are brittle, and their spleens are unusually big. The female mice produce fewer and smaller litters than normal mice. They also are more likely to develop high blood pressure and a disease that resembles human leukemia. In fact, problems of one sort or another afflict almost every major organ system in their fragile, overweight bodies.
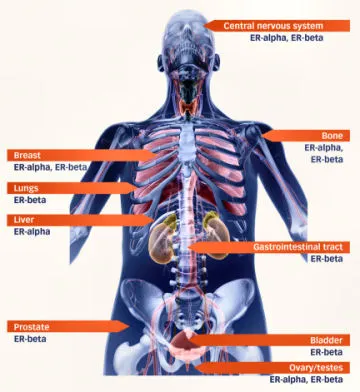

What these mice lack is the gene for an important molecule needed to fully respond to the hormone estrogen. Known as estrogen receptor beta (ERb), this molecule mediates most of the effects of estrogen not traditionally associated with the hormone. By genetically engineering both male and female mice without the receptor, researchers are digging up clues to its many important roles in people.
Discovered only a decade ago, the beta receptor has been found to protect against cancer, keep the immune system in check, help serious trauma patients survive their injuries, and keep people from being too anxious. A recent spate of studies on the receptor could lead to a new generation of hormone-based drugs for infertility, breast cancer, irritable bowel syndrome, depression, and a myriad of other conditions.
“Estrogen receptor beta, in particular, is involved in many, many tissues. It shows that estrogens are extremely important not only in reproduction, which everybody knows, but in other aspects of health as well,” says Gustafsson, of the Karolinska Institute in Stockholm, Sweden.
Estrogen, a hormone that circulates in the blood, comes mainly from the ovaries, but is also made in small quantities by the placentas of pregnant women, the liver, adrenal glands, and breasts. In males, certain cells in the testes produce low levels of estrogen.
Estrogen affects various organs by entering cells and attaching to receptors inside them. Stimulated by estrogen, the receptors activate genes that change how the cells behave. But in every organ system, the changes may be different.
Understanding estrogen’s effects has been complicated by scientists’ long-held assumption that the hormone bound to only one type of receptor, now called estrogen receptor alpha (ERa). The uterus boasts a high concentration of the alpha receptor, and since researchers thought estrogen was only important for female reproduction, they had no reason to go looking for a different receptor in other organs.
But in 1996, while searching broadly for new hormone receptors, Gustafsson stumbled on estrogen receptor beta. That finding turned the field of estrogen research around, says Kenneth S. Korach of the National Institutes of Health.
At the time, Korach was studying mice missing the alpha receptor. It had been a surprise that these mice could even survive. “The prevailing view was that you needed to have estrogen and you needed to have estrogen receptors,” he says.
The discovery that there was a second receptor seemed to explain his results. If the mice missing the alpha receptor still had a functional beta version, their bodies still could respond to estrogen. At first, “everybody thought alpha was covering for beta and beta was covering for alpha,” says Korach.
But when Korach bred mice lacking both receptors, the new mice survived too. “That clearly indicated that one does not need to have estrogen-receptor function to live,” he says, “or for any type of developmental biology.”
Though mice without the beta receptor can live, they are plagued by a list of health problems—a list that continues to grow as researchers observe the mice aging.
Bonnie Deroo, of the University of Western Ontario, in London (Canada), studies one impact of the missing beta receptor in female mice-fertility problems. Though these engineered mice can produce offspring, their litters are few and small. “Understanding how estrogen regulates ovulation may reveal causes of infertility in humans,” she says.
Deroo’s research focuses on estrogen in the ovaries, the female egg-producing organs nestled above the uterus. Ovaries, Deroo has found, have both alpha and beta estrogen receptors, but not in the same cells.
Deroo thinks the beta receptor may be involved in ovulation, the monthly release of an egg into a woman’s uterus. Normally, estrogen produced by the ovaries causes the pituitary gland to release another hormone, called luteinizing hormone (LH) that directly triggers egg release. Deroo is trying to figure out what part of this cascade of hormones malfunctions in mice lacking beta receptors.
Korach studies this process to help women with infertility problems too, but he also points out another potential payoff from the research: estrogen receptor beta could be blocked in women as a form of birth control.
Cancer watchdog
Produced by plants, estrogen-like compounds called phytoestrogens are found in foods including soy and coffee. Some scientists have speculated that phytoestrogens played a key role in the lower incidence of prostate and breast cancers found for many years in Asian populations with soy-based diets.
This link doesn’t surprise Gustafsson—the beta receptor responds to phytoestrogens much more potently than the alpha receptor does. Gustafsson thinks the beta version holds promise in combating cancer.
When estrogen binds to an alpha receptor, it usually signals the cell to start copying itself. This response is important in many situations, including making women’s breasts and uteruses grow during pregnancy. But if something goes wrong while the estrogen receptor alpha is signaling the cell to multiply, cells may begin to grow uncontrollably and cancer could result.
“ERa seems to be, in that context, the bad boy or the bad girl,” says Gustafsson. “But ERb is the watchdog.”
Gustafsson’s lab has shown that compounds that bind to only the beta receptor stop cells from multiplying. In mice, these drugs impede the growth of breast and prostate cancers.
Other labs studying the role of estrogen receptors in relation to cancer have found similar links in both males and females. Healthy breast and prostate tissues have more beta receptors than breast and prostate cancer samples, numerous studies have shown. Scientists have hypothesized that the lack of beta receptors in the cancer tissues allows the cancer to grow in the first place.
Compounds that bind to the beta receptor but not the alpha version, called ERb-specific agonists, also may be beneficial for a host of other health problems. These drugs, which turn on the beta receptor just as estrogen would, could even help patients survive severe physical injuries.
A paper published in the journal Shock in August 2000 found that females are more likely to survive traumatic injury, and less likely to go into sepsis—a severe bodily reaction to infection—than males. Scientists hypothesized that this finding could be because females have more circulating estrogen than males.
To follow up on this finding, another group of researchers gave estrogen or ERb-specific agonist to mice with lung injuries. In both groups of mice, the levels of inflammatory compounds in the blood instantly decreased.
The team, led by Heather Harris of Wyeth Research in Collegeville, Pa., also studied rats bred to develop inflammation in the joints, intestine, and skin. Scientists use these rats as models for human inflammatory diseases like rheumatoid arthritis and irritable bowel syndrome. Ten different ERb-specific agonists given to the afflicted rats decreased the inflammation and joint swelling.
Around the same time the link to chronic inflammation was found, researchers studying mice lacking the beta receptor noticed that the mice’s immune systems weren’t good at fighting off pathogens.
When the Wyeth scientists gave an ERb-specific agonist to mice infected with bacteria, more than 80 percent survived. No mice without the drug survived the infection.
Lessening the pain
For people with chronic diseases, ERb-specific agonists could also decrease pain, says Gustafsson. While no such pain drugs have been developed yet, clinical observations of women depleted of estrogen show a possible link between the hormone and pain levels.
One such observation comes from women being treated for breast cancer with aromatase inhibitors, drugs that stop the body from making estrogen. This means that there’s no, or very little, estrogen to tell the alpha receptor to make cells multiply. But it also means there’s no estrogen to bind to the beta receptor.
Aromatase inhibitor side effects include osteoporosis, which is thought to involve estrogen receptor alpha. But another side effect surprised doctors: Women taking aromatase inhibitors, such as Arimidex, reported feeling intense pain all over their bodies.
Now, based on experiments with mice, Gustafsson thinks the pain is linked to estrogen receptor beta.
“It seems that, generally speaking, ERb increases the pain threshold, that is, it decreases pain,” says Gustafsson, “and ERa decreases the threshold, so everything hurts.”
Put together, he says, the findings on the beta receptor’s link to inflammation and pain mean that the next ibuprofen-like painkiller may be an ERb-specific agonist.
Based on his mice observations, Gustafsson thinks similar drugs could be designed as new anti-anxiety or anti-depression drugs.
When women reach menopause around age 50, and the amount of estrogen in their bodies sharply falls, many suffer severe depression. Gustafsson thinks this is no coincidence.
Estrogen receptor beta, after all, is plentiful in the brain. As embryos, mice develop beta receptors in their brains by the 12th day of growth. And adult mice lacking the receptor are violent and anxious, says Gustafsson.
“These mice attack each other,” he describes. “They take the fur off each other.”
In 2005, researchers led by Robert J. Handa of Colorado State University in Fort Collins performed a number of anxiety-related tests on rats treated with either ERb-specific agonists or ERa-specific agonists. When alpha was stimulated, the researchers found, the mice became more anxious, avoiding open areas in a maze, for example. The rats given ERb-specific agonists, however, showed none of these anxious behaviors.
From the moment he discovered the second estrogen receptor, Gustafsson says, he knew its function wouldn’t be the same as the original receptor. After all, he’d isolated it from a rat’s prostate, and estrogen hadn’t been thought to have a function in the prostate. A decade of findings on beta receptors has strengthened the view that the second receptor has little overlap with the alpha version. It’s also become clear just how many body functions the beta receptor mediates. The next challenge will be to find ERb-specific compounds that can bind to the receptor in certain organs but not others.
Hormonal origins
The evolutionary history of estrogen receptors may explain their wide range of functions. In 2003, a scientist studying mollusks discovered that these shelled organisms have only one hormone receptor (SN: 9/20/2003, p. 180), and it binds estrogen. When the scientist, Joseph Thornton of the University of Oregon in Eugene, analyzed the receptor further, he found that it’s like a hybrid of the two human estrogen receptors. The discovery suggests that estrogen receptors must have been around early in evolution, before invertebrates and vertebrates diverged.
Gustafsson hypothesizes that this primitive receptor gave rise to the two estrogen receptors and, eventually, perhaps to more modern steroid receptors, including the glucocorticoid, vitamin D, and progesterone receptors.
“One can easily envisage that the estrogen receptors, being the first steroid receptors, must have had significant functions in regulating lots of systems in these early primitive organisms,” he says.







