Some North American bats leave hibernation with little sign of the devastating effects of white-nose syndrome, a fungal epidemic that has claimed the lives of some 5 million bats since it first emerged in winter 2005. But even though those bats seem to have survived the fungal disease, their immune systems reactivate and can then inexplicably — and devastatingly — kick into overdrive, a new study reports.
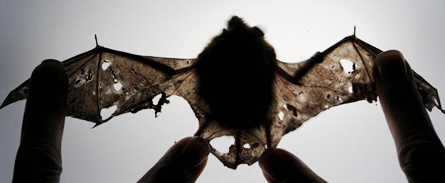
These animals appear to have what immunologists call immune reconstitution inflammatory syndrome, or IRIS. The discovery of the condition in bats “is the first potential example of IRIS that has ever been seen outside a human patient,” observes wildlife pathologist Carol Meteyer of the U.S. Geological Survey in Madison, Wis. Until now, most IRIS victims have been HIV patients treated with medicines to restore flagging immune systems.
Together with Daniel Barber and Judith Mandl at the National Institute of Allergy and Infectious Diseases in Bethesda, Md., Meteyer describes the bats’ IRIS-like disease in the Nov. 15 Virulence.
Animals afflicted with white-nose syndrome acquire the infection during hibernation, when their bodies chill to near-freezing and their immune systems effectively go into suspended animation. Despite its name, the disease appears to kill its hosts by eroding large patches of tissue in the animals’ wings. Geomyces destructans, the infecting fungus (SN: 9/10/11, p. 22), then begins filling in those divots with its own cells, Meteyer says. Stricken bats become weak, dehydrated — and eventually become unable to fly.
IRIS seems to shows up in bats that have largely evaded this scenario. Instead, these animals emerge from hibernation with normal-looking wings. But as their body temperature warms back up and their immune systems reactivate, their health takes a nosedive. Within days, dark patches riddle their wings. The patches point to where immune cells known as neutrophils have begun unleashing an inflammatory assault against the fungus they’ve been summoned to eradicate. Over the next two weeks, these and other immune cells encapsulate the fungal patches, walling them inside scablike structures. Soon the scabs fall away, leaving the wings with huge holes. Flight becomes limited, if not impossible.
The first signs of this unusual condition emerged in May 2008, Meteyer recalls: “I was being sent bats found in front yards. They could not fly.” She quickly dismissed her initial suspicion, rabies, after examining the first animal’s wings. “Under microscopy, I could see the wings were tattered with holes.” Further probing would link inflammation to this damage.
Animals exhibiting IRIS can be nursed back to health in the lab with food, drink and warmth, Meteyer has shown (SN Online: 10/26/11). But in the wild, it’s doubtful such animals stand a chance, she says. If the fungal disease doesn’t kill them outright, IRIS will. “They just face this double whammy.”
What her team describes in its new paper reflects what appears to happen in HIV patients, says Samuel Shelburne, an infectious diseases physician at the University of Texas M.D. Anderson Cancer Center in Houston. Although HIV depresses their immune systems, these patients can acquire other types of infections — such as from bacteria — that they initially don’t fight. But then when drug therapy reconstitutes that immunity, these people can develop an overwhelming inflammatory response.
Bats might therefore offer an animal system in which this situation develops, he says, so that researchers can probe why only some HIV patients develop the devastating IRIS backlash.
In fact, Meteyer notes, her colleagues at NIH hope to test just that using colonies of hibernating Mexican free-tail bats. This species, unlike those naturally affected by white-nose syndrome, survives well in captivity and is not endangered.






