The first living laser is nothing to be afraid of. It’s just a single cell pulsing with light that may lead to new ways of probing microscopic realms.
And maybe — just maybe — laser bunnies.
The secret to the cell’s splendor is called green fluorescent protein, or GFP, researchers report online June 12 in Nature Photonics.
First discovered in jellyfish, this glowing protein has long helped biologists illuminate cells and their inner workings. When struck by blue light, its electrons essentially jump up and down and fluoresce with green light.
The green light emitted by one GFP molecule can also trigger another GFP molecule to spit out identical green light. Two physicists have now used this phenomenon, called “stimulated emission,” to set off the chain reactions required to make laser light from GFP.
“They’ve shown that you can do lasing action in a live cell without destroying the cell,” says Stefan Hell, a physicist at the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany. He has used stimulated emission to create microscopes with record-breaking resolution.
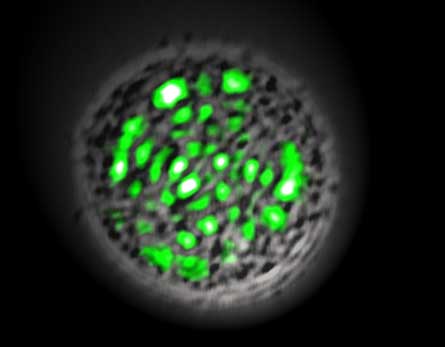
For their first experiment, Seok-Hyun Yun and Malte Gather of the Wellman Center for Photomedicine at Massachusetts General Hospital and Harvard Medical School placed two mirrors close together and filled the space between with liquid containing GFP. Brief pulses of blue light excited the proteins — which set each other off with help from the mirrors and created pulses of green laser light more intense than the original blue light.
The principle is similar to that of dye lasers, first invented in 1966. Dye lasers can also amplify light using dissolved organic molecules — not proteins, but compounds such as coumarin dyes, which are derived from substances in grasses that smell like freshly mowed hay.
Then the physicists pimped their GFP laser, replacing the free-floating molecules with a single mammalian cell containing a bit of DNA that churns out a mutant form of the fluorescent protein. When stimulated, the cell could pulse with light a few hundred times before the GFP gave out — or in technical terms, photobleached.
“The interesting thing here is that the cell keeps making the GFP protein as it grows,” says Steve Meech, a physical chemist at the University of East Anglia in Norwich, England. Unlike a dye laser, a living laser should be able to heal itself over time by replacing photobleached molecules.
Other colors should also be possible.
“There are other types of fluorescent proteins found in coral reefs in the sea that might be useful for lasers,” says Yun. “We’re currently testing several of those.”
Lasing proteins could provide a more sensitive biological tag for identifying molecules inside cells. Changes in the distribution of the proteins within a cell should change the characteristics of the emerging laser light.
Yun is also trying to teach groups of cells to lase together and to replace the mirrors with reflective nanostructures inside cells.
“If that’s possible, you might be able to make cells lase without any external help,” says Hui Cao, a physicist who builds biologically inspired lasers at Yale.
Cellular lasers embedded in tissue could be part of a new kind of imaging technology. Or maybe someone creative will eventually one-up scientists who have used GFP to make glow-in-the-dark rabbits — by creating a laser bunny.





