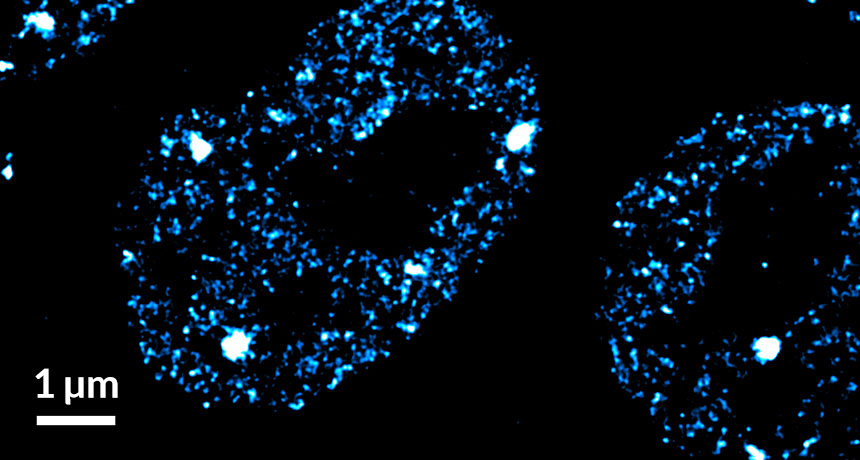
MARKING THE SPOTS In this super-resolution microscope image of mouse embryonic stem cells, molecules of the enzyme that copies DNA instructions into RNA messages cluster in multiple spots within the nucleus. White spots have more of the enzyme, RNA polymerase II, concentrated in them than in the cooler blue spots.
W.-K. Cho et al/Science 2018