Life from scratch
Relaunching biology from the beginning
A short stroll from Boston’s Charles River, behind a sheath of blue glass on the seventh floor of a Harvard Medical School research building, Jack Szostak is getting set to replay the greatest event on Earth.
He and his 15-member team of graduate students and young postdoctoral research fellows are well on their way to starting biology from scratch — more than 3.5 billion years after it first emerged.
The feat would qualify as creation of life in a test tube if it weren’t for one thing: Szostak’s lab does not rely much on test tubes. “I know exactly where it will happen,” said postdoc Alonso Ricardo, from Cali, Colombia. It will most likely be in a 1.5-milliliter tapered plastic centrifuge tube “smaller than my little finger.” And unlike the first time — when life formed on its own — the second time it will get a boost from human ingenuity and the lab’s elaborate organic chemistry equipment.
Szostak’s endeavor is very different from another artificial life project led by biologist and entrepreneur J. Craig Venter. Venter’s team is using chemical sequencing machines to make a panoply of genes for the highly evolved parts of a modern microbe. Recently he and his colleagues announced that they had inserted an entire genetic blueprint, modeled on a known microbe but built from scratch, into a microbe of another species where the synthesized DNA took over (SN: 6/19/10, p. 5). Venter’s ultimate aim is to build designer organisms with novel and fully contemporary genomes.
Szostak has a far more fundamental aim: to show how unguided natural events might have led to life on Earth in the first place, and to show how the scenario might also play out in myriad other places in the universe. Like bookends on a long row of volumes, the two exercises would frame the story of evolution so far.
In his neat corner office outside the rows of lab benches and work bays, the 57-year-old biochemist leaned forward and explained a deep motivation: “What we’d like to see is, from initial chaos and randomness, how something useful emerges. What we are trying to do, to understand, is how Darwinian evolution can emerge from chemistry…. If we can get a self-propagating chemical system that can evolve, yeah, I’d call that life.”
A place to start
Szostak brings a lot of tools to the project. He has already made his mark on biology throughout a career puzzling over and exploring the workings of DNA and its cousin, RNA. He was a winner last October of the Nobel Prize in physiology or medicine, along with Elizabeth Blackburn of the University of California, San Francisco and Carol Greider of Johns Hopkins University School of Medicine in Baltimore. In the 1980s they showed how telomeres, distinctive caps on the ends of chromosomes, protect a living cell’s DNA and genes from degradation.
Szostak, a U.S. citizen now, was born in London, where his father was stationed with the Royal Canadian Air Force. After returning home, Szostak enrolled in McGill University in Montreal at age 16. At 19 he took his degree in cell biology to graduate school at Cornell University. He dove into genetics. Nature published an extract from his biochemistry Ph.D. thesis — on synthetic RNA. At 26 he joined Harvard’s faculty, where he is now a professor of genetics and a Howard Hughes Medical Institute investigator.
By the mid-1980s, electrifying word swept the field of DNA and RNA research. Tom Cech of the University of Colorado at Boulder and Sidney Altman of Yale University independently discovered that RNA — believed to be a mere messenger, carrying genetic blueprints from DNA-based genes to cellular machinery for making proteins — had another trick. It could fold into complex shapes, forming an enzyme that vastly speeds up the natural rate of some reactions (SN: 11/27/82, p. 342). Until then, the only known enzymes were specialized proteins. How life had first made proteins without enzymes, which presumably had to be proteins themselves, had been a chicken-and-egg conundrum.
But the discovery that RNA can be an enzyme, dubbed a ribozyme, potentially able to boost its own replication, led to fresh ideas about how life might have started. Cech imagines an early “RNA world,” where life could have taken its first steps while ignoring all the amino acids floating in the prebiotic soup. Only later, with ribozymes as tools, need life have developed a genetic code for stringing amino acids into proteins — gradually turning metabolic duties over to those proteins.
Ten years ago Szostak and Pier Luigi Luisi, then at the Swiss Federal Institute of Technology in Zurich, settled an argument by writing a paper together. They had been debating the order of important steps in the origin of life. Luigi Luisi favored the membrane first: It would hold a primitive “protocell” together and keep its vital chemicals concentrated. Szostak figured that genetic machinery able to copy itself and assure that its features get passed from generation to generation was paramount. The team’s paper, “Synthesizing Life,” written with David P. Bartel of MIT’s Whitehead Institute for Biomedical Research, came out in Nature in early 2001. It split the difference, arguing that life would need both at the start: a whole cell. Its second paragraph asked “How simple can a cell be and still be considered as living?”
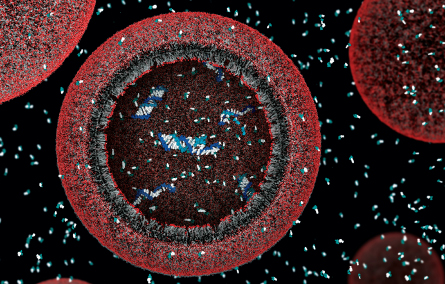
The researchers conceptually stripped it to almost nothing. On the outside, a uniform membrane of simple and ubiquitous lipid, or fat, molecules. Inside, a few strands of nucleotides, some possibly folded into ribozymes.
“After we wrote that paper I figured, well, we put these two ideas together, so we’d better do some experiments,” Szostak recalled.
Szostak and his team already have a stripped-down system for making tiny hollow spheres, or vesicles, from simple lipid membranes. And lean and mean genetic material is available from simplified versions of today’s RNA. The researchers’ molecules are showing signs of being able to assemble, replicate, mutate, and be led by the power of natural selection toward complex metabolism.
Such work has helped put Boston and Cambridge among the elite centers of studies of life’s origin, and Szostak is now devoted essentially full-time to synthesis of life. “They won’t get it done in 2010, but with some good luck and a lot of work it could in the next year or two,” says Gerald Joyce, an RNA specialist at the Scripps Research Institute near San Diego, a traditional powerhouse for origins work. Joyce’s specialty is the creation of ribozyme systems that replicate and even evolve somewhat — on the verge of life, but not quite there. Of Szostak’s work, he says: “They have some really hot chemistry going there. If they can rev it up some more, it — life — will just start working. Once you light Darwinian evolution, it takes off.” Others guess it may be five or 10 more years. Szostak won’t play that game but says, “How old am I? I think I have time.”
Szostak also doesn’t care for guessing exactly how life happened the very first time and then duplicating it. Once one system works, he figures the experience will generate clues to other ways. And he doesn’t think his work will settle an old argument among pioneers in the field: At what locale did life first take root? “I think it might have taken them all,” he says. These include a “warm little pond” as Charles Darwin proposed, or the oceanic prebiotic soup suggested independently by Aleksandr Oparin and J.B.S. Haldane in the 1920s, or deep-sea hydrothermal vents, or wave-washed beaches, or lightning-lashed volcanoes, or terrestrial hot springs or other hot spots.
Protolife plays out
Wherever life first flickered into being, it may have moved rapidly to occupy a wide range of environments. In a paper published in April in Annual Review of Biophysics, Szostak and his graduate student Itay Budin argue that once even the crudest living system appears, “there is a nearly limitless set of plausible adaptations that would be beneficial for early cells.” In a recent speech in Florida at a meeting of the Lasker Foundation, which awarded Szostak, Blackburn and Greider its annual prize for basic medical research in 2006 (an example of why Laskers are tips to future Nobels), Szostak said he expects to see firsthand “an evolutionary arms race” among his first protocells as they compete and evolve ever-superior tools for survival.
For decades, science has known that raw materials for life were surely abundant on early Earth. In a famous 1953 experiment at the University of Chicago, chemist Harold Urey and, chiefly, his graduate student Stanley Miller showed that a primitive atmosphere of methane, ammonia, water vapor and hydrogen, when blitzed by a lightninglike spark, produced a residue rich in amino acids — building blocks of proteins. The demonstration set off a wave of experiments showing that other conditions can do it too. And not only here. Outer space also seems loaded with the Lego blocks of life. Meteorites can contain thousands of organic molecules, including amino acids.
Further and vital reassurance to Szostak’s assumption that the needed chemistry was readily available to the first life came early last year. At the University of Manchester in England, chemist John Sutherland and his team reported discovering a natural set of reactions by which ribonucleotides, the fundamental units of RNA, could have built up on the young, lifeless Earth.
“It’s almost impossible not to make amino acids, sugars, lipids, alcohols and almost anything you can think of,” says geochemist Robert Hazen of the Carnegie Institution for Science in Washington, D.C. “There is almost too much stuff, too many possibilities, to narrow it down to just one idea.”
What is firmly supposed is that somehow, somewhere beyond 3.5 billion years ago, before the first signs of microbial life appear in the fossil record, and after about 4.2 billion years ago, when the planet had cooled and became habitable, a vesicle wrapped itself around a few pieces of chained nucleotides. Those nucleotides hooked up into strands as more seeped inside. And, by higgledy-piggledy chance after uncounted similar episodes that went nowhere, some of these innards took the form of a ribozyme with the power to speed reactions, including replication. The protocell divided. It carried the protogenetic material into further generations, the occasional copying mistake opening the door to evolution.
These steps are the ones Szostak’s team hopes to watch unfold. The strategy is simple — find molecules that can do the necessary replication quickly. Executing it is difficult — nature had millions of years and an enormous number of ponds, springs, beaches and oceanic hot spring vents to work with. “This is a molecular biology project, but as a practical matter we are almost a full-time organic chemistry synthesis operation,” Szostak says.
The project’s achievements so far can be broken down in terms of a few larger tasks:
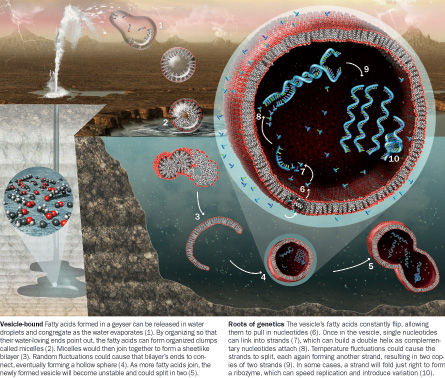
THE MEMBRANE: Modern cell membranes are tough, and highly protective. Bristling with receptors and gates, these layers don’t let just anything leak in or out. Their bricks and mortar are phospholipid molecules — so named because of a prominent phosphorus group that provides stability and strength. One end of each molecule is attracted and the other repelled by water molecules. This split personality leads them to form roughly spherical, closed membranes in two layers, with the water-friendly ends all facing the exterior and the water-shy ends tucked along the membrane’s interior seam.
Because simpler membranes are more likely to have formed from the natural organic chemistry factory on prelife Earth, the protocell project is working on plainer lipids — chiefly ordinary fatty acids. Like phospholipids, these simpler fatty acids form double-layered protocell walls that keep large molecules in. But unlike phospholipids, they allow nucleotides, the small building blocks of genetic material, to pass through.
THE GENETICS: Making a system that can duplicate itself without modern life’s elaborate protein enzymes is the project’s biggest hurdle, but progress has been dramatic.
DNA and RNA are made of nucleotides. Each unit is a three-part molecule with a phosphate at one end, a sugar in the middle and a variable nucleobase at the other end. The nucleobases come in various sorts — providing the “letters” of the genetic code and also providing the chemical bonds that let one DNA strand cling to another to form the famed double helix. Szostak’s team plans to get a random RNA or similar molecule in the vesicles, let more nucleotides enter one by one and, by linking to the first one, produce two linked strands of an RNA-like molecule. Temperature oscillations or chemical changes should cause complete double helices to come apart, each then rebuilding the whole as fresh nucleotides link on.
But with normal RNA this process is too slow. Ricardo is experimenting with closely related, more reactive and reliable nucleotide systems. “We just tweak the RNA here and there, change a few atoms,” Ricardo says.
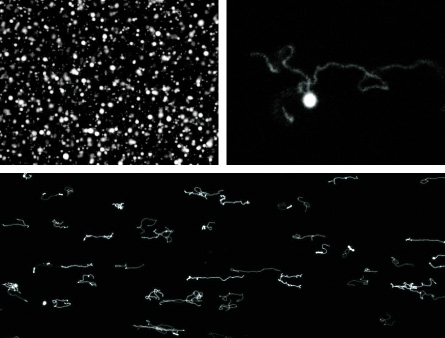
One called TNA replaces RNA’s ribose sugar with a sugar called threose. Another variant is called morpholino NA. This list goes on, with many more to try. The self-assembly and replication is going better but has not yet been able to reliably and speedily duplicate lengths of genetic material long enough — many dozens of nucleotide units — to fold into long polymers. But the strands are getting longer all the time.
GROWTH AND EVOLUTION: Graduate student Ting Zhu, a native of Shanghai, is the lab’s vesicle man. When he joined the group a few years ago the fatty acid lipid vesicles were already standard. Szostak wanted to know how to make solutions of same-size vesicles and to see how fast they could grow. Zhu, a mechanical engineering graduate from MIT, first invented a tiny sorting machine. Szostak suggested he study in detail how the membrane grows by incorporating new fatty acids, a step at a time.
Zhu had a simpler idea: He dumped a high concentration of micelles — small balls of huddled fatty acids — into a suspension of vesicles. He put them in the lab’s Nikon TE2000S inverted epifluorescence microscope — a device big enough to fill a closet —and watched a most remarkable thing happen. The vesicles writhed. They grew what looked like wriggling hair. They eventually transformed themselves from round balls into long filaments. Their membranes had grown so fast that they couldn’t absorb fluid quickly enough to stay plumped up.
Soon Zhu discovered that simple shaking of the fluid broke the filaments into a new generation of vesicles without spilling their interior contents — and he has shown it can be done while the vesicles are full of nucleotides.
Further work has revealed that simple alcohol buffers can cause each filament to form what looks like a string of pearls that then floats free, forming a new generation. It became clear that the knotty problem of getting protocells to divide might have a simple enough answer: a stew of raw materials to transform the cells into long cylinders and changing turbulence or chemical conditions to split those filaments.
The team sees two ways, if the above steps work, to witness selective, Darwinian pressure alter protocell behavior. For only if the protocells can evolve might they merit status as living cells.
Among discoveries in the lab over recent years is that when a vesicle’s cargo of nucleic acid polymers fills up to the point it strains the cell’s wall, the membrane will greedily absorb any fatty acids floating by. Cells whose interiors grow the fastest then should, in competition with others, take the lion’s share of available lipids, eventually eliminating their competition. Thus, any random changes in the interior, fledgling ribozymes that could speed up nucleic acid replication rates will spread rapidly through an evolving population.
Alternatively, another Darwinian tableau might see the random development of ribozymes able to add phospholipids, the stuff of modern membranes, to outer fatty acid membranes. The result would be more stable structures superior to other cells, also able to hang on more tenaciously to any fatty acids they acquire. Again, those with the most advanced membranes would dominate — up to a point.
This scenario, Szostak says, has a twist. If the phospholipid proportion rises too high, it will prevent small nucleotides from getting inside, starving the genetic machinery of new materials.
The solution for the cell is to find a way to put gates or pores in the membrane that allow fresh nucleotides in. How that might happen is sheer speculation. But Szostak is sure that, given enough time and enough trials, protocells made with his general formula will almost certainly evolve new characteristics that give them an edge over others. And when that happens, Szostak may finally see how, from initial chaos and randomness, something useful emerges.