Like teenagers, cells require constant communication with their peers. Today’s teens chatter endlessly over wireless networks. Cells, on the other hand, seem a bit more old-fashioned. A clandestine web of high-speed wires physically links cells like a biological Internet, scientists have discovered.
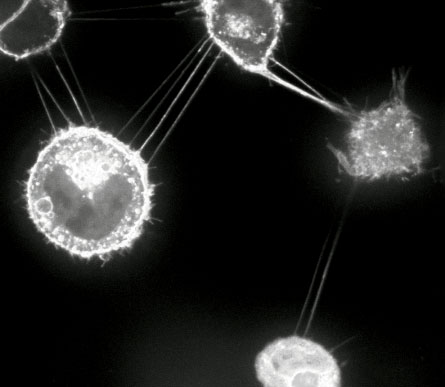

These long, filamentous fibers are called tunneling nanotubes. They lurk in lab dishes of human kidney cells, immune cells and cancer cells. The tunnels share the same tiny dimensions as the nanotubes that chemists create with carbon. But these nanotubes aren’t built by scientists. Tunneling nanotubes grow with no external interference, and they seem to offer a heretofore unknown way for cells to communicate.
“These structures have been there all along. If you don’t know what you’re looking for, you miss many things,” says Hans-Hermann Gerdes, a cell biologist at the University of Bergen in Norway who reported the first official sighting of the tiny fibers in 2004.
Since then, a flurry of work has begun to uncover the prevalence and purposes of these conduits between cells. The covert, long-range tunnels could explain developmental mysteries like how individual cells coordinate growth into complex tissues and how immune cells streamline efforts to rapidly fight off intruders. Living nanotubes can also shuttle organelles, including the energy-producing mitochondria, between cells, a study published in December found.
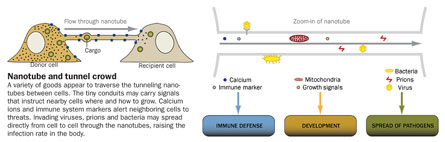
Such fast, direct communication lines can also ferry unwanted guests. Just as fast Internet connections spread computer viruses, tunneling nanotubes can carry dangerous cargo. Nanotubes can be commandeered by unfriendly forces to spread disease: Recent studies show that bacteria, viruses and infectious prion proteins can all move through the nanotubes for nefarious purposes.
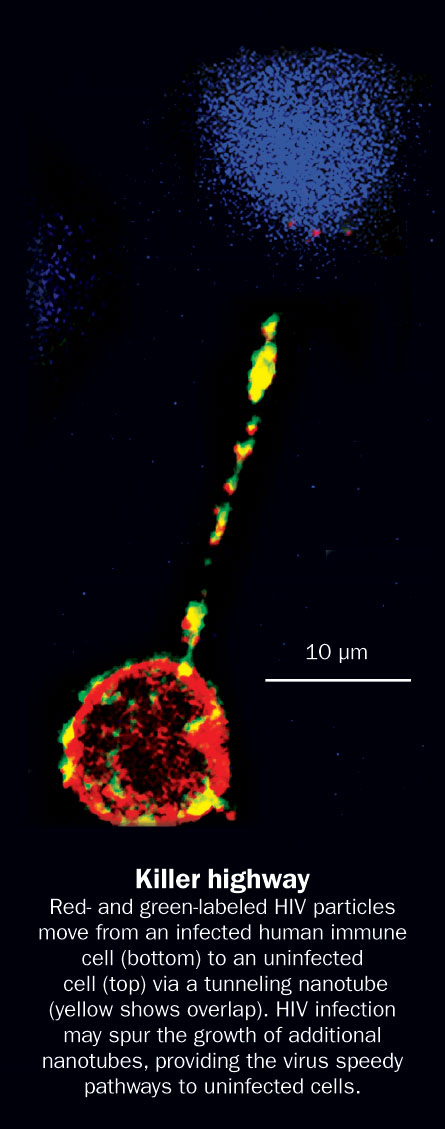
In one recent study, Daniel Davis, an immunologist at Imperial College London, saw glowing HIV particles creeping from an infected immune cell to an uninfected cell through the taut nanotube wires. A different study published in January found that human cells build more networking nanotubes when infected with HIV, providing a scary scenario for ramped-up virus transmission. And recent data suggest that deadly prions—infectious, misfolded proteins that cause brain-wasting diseases like Creutzfeldt-Jakob in humans and mad cow in cattle—can travel through tunneling nanotubes to infect healthy brain cells.
To date, most nanotubes have been found on cells grown in lab dishes. And, critics say, cells plopped down in a dish, away from the normal biological context, probably act a little strange, like hermits who have been away from civilization for too long. Ultimately, these scientists say, whether nanotubes carry harm or good to other cells in lab experiments may be irrelevant to the body.
“This area is very new,” says Walther Mothes, a cell biologist at Yale University who questions nanotubes’ significance for disease. Currently, more reviews on tunneling nanotubes exist than research papers, he points out. “We need more evidence.” Such evidence has come slowly. To study and evaluate the importance of nanotubes in natural systems, scientists first had to find them.
The jungle in there
Tunneling nanotubes thread among cellular debris and other kinds of cell connections, making them hard to spot—like trying to pick out a piece of floss from a tangled bird nest. Looking through a microscope at a group of cells, “a jungle of extracellular structures” meets the eyes, Gerdes says.
An even greater obstacle to finding nanotubes was their frailty. These wires are like the ultrafine, rigid glass tendrils that remain after a glassblower stretches a vase. They readily break under mechanical stresses, the kinds of jostles and bumps that take place when preparing a cell sample for the microscope. Common fixatives like formaldehyde, used for preserving cells before imaging, obliterate nanotubes. Even light can destroy them—exposed to light, nanotubes start to vibrate, ultimately shattering. Better, more precise laser microscopes and the development of sophisticated imaging techniques finally illuminated these elusive structures.
Once tunneling nanotubes were spotted, scientists saw many different kinds. “If you look down a microscope at cells, lots of different cells have these connections, and they may be very different,” Davis says.
Researchers led by Eliseo Eugenin at Albert Einstein College of Medicine in New York City recently found that human cells isolated from blood can form two distinct kinds of tunneling nanotubes: Long thin ones and short wide ones. The “long” tubes can extend 150 micrometers in length (about one and a half times the width of a human hair—quite a distance for a small cell). The short ones extend about 30 micrometers.
Nanotubes don’t all look the same. They don’t all form the same way, either. So far, they have been shown to develop in at least two ways—by growing out from one cell to contact another or by remaining linked as two cells move apart.
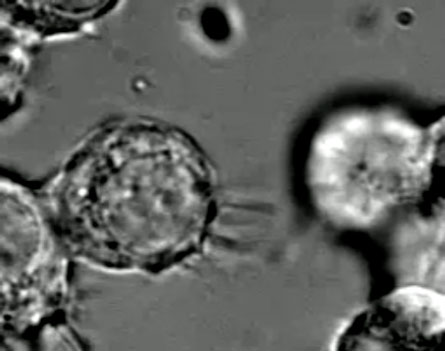
Scientists including Gerdes have seen the nanotubes form. Over the course of four minutes, he and his colleagues watched a stationary rat cell, alive on a microscope dish, extend several spindly protrusions toward another stationary cell, like fingers groping around in the dark. One feeler made contact with the other cell, and a nanotube was formed, while the rest of the feelers retracted into the cell body.
“They are really dynamic,” Eugenin notes. “The formation can happen in minutes.”
Some tunneling nanotubes develop only after contact with other cells, like thin ribbons of caramel left when two candied apples are pulled apart. In the case of the human immune cells that Davis’ group studied, the tunnels formed when two cells bumped into each other. As the cells spread apart, a wire-thin nanotube stretched between them. The link is created in just seven minutes.
Davis’ team found that immune cells produce significantly more nanotubes when the cells touch for more than four minutes. In contrast, cells that touch for less than three minutes rarely have nanotubes, suggesting that a brief bump together is not enough time.
Researchers think that the differences in tunneling nanotubes and how they form probably reflect varying needs of cells to send and receive information. Immune cells may coordinate a speedy counterattack by using nanotubes to quickly exchange calcium—a signal the body uses to warn other cells of harmful invaders. Proteins that tell developing cells when and where to grow may also flow through nanotubes. Such proteins could direct adjacent cells to grow in such a way as to create the complex patterning seen in the fruit fly wing, for example. Nanotubes may also carry a yet unknown heart development signal: They have been observed connecting naïve, unspecialized cells to mature heart cells, hinting at a new way cells may find their adult identity.
Nanotubes can also carry organelles, including the energy-producing mitochondria and the endoplasmic reticula, which house the cell’s protein factories, Gerdes and his colleagues showed in a paper published December 10 in Experimental Cell Research. Since mitochondria contain their own DNA, cells could even be swapping genetic material through these tunnels.
Such a free exchange of mitochondrial DNA among nondividing cells could challenge current understanding of cells as separate functional units, Eugenin says. “What you read in books that cells are discrete entities, that’s not true anymore. If you see this in a broad way, it’s a huge, huge, huge change from how we think about cells.”
Dangerous cargo
Conduits that carry useful information can also transport danger, in the form of viruses or harmful prion proteins, from cell to cell. The tubes may explain a conundrum involving HIV, for example.
Researchers have shown that HIV, the virus that causes AIDS, is about a hundred times more likely to infect cells in contact with each other—as cells exist in the body—than a cell grown in isolation, as in lab dishes. If the virus is washed over the outside of a solitary cell, the cell can fight off the infection with antibodies. But in the body’s close-knit community of cells, the virus spreads like wildfire.
Scientists have been puzzled by the ease with which HIV can spread from infected cells to uninfected cells in these communities, even in the presence of neutralizing antibodies. Tunneling nanotubes offer one explanation for this phenomenon. HIV may move underground.
“The virus is hijacking the system,” Eugenin says. “The pathogens are using and abusing the cell’s own machinery.”
Davis’ research group tagged HIV particles with green fluorescent protein and then infected human immune cells. Using highly sensitive microscopes, the researchers watched as green globs of HIV made their way along a tunneling nanotube and into an uninfected cell. This new mode of virus transfer is “rapid, long-distance intercellular transmission,” the researchers concluded in work that appeared last year in Nature Cell Biology.
In a separate set of experiments, Eugenin’s team used HIV to infect blood cells from healthy volunteers. After a day, more of the infected cells had sprouted tunneling nanotubes than had uninfected control cells. The results suggest that once inside a cell, HIV tricks cells into making more tunneling nanotubes, through which the virus can travel and infect more cells, Eugenin and colleagues reported in the January 8 issue of Cellular Immunology.
Viruses aren’t the only dangerous goods to travel through tunneling nano-tubes. A paper online February 8 in Nature Cell Biology describes how brain-wasting prions use tunneling nanotubes to travel from an infected neuron to an uninfected neuron. What’s more, nano-tubes may explain the mystery of how prions move from the intestinal system, often the site of an initial infection, to the brain. In this new study, Chiara Zurzolo of the Institut Pasteur in Paris and her colleagues show how prion-infected immune cells, which can circulate in the blood from gut to brain, can form nanotube connections to brain cells. If this happens in the body as well, it may lead to prion infection in the brain.
Gerdes speculates that figuring out a way to manipulate nanotube connections might reduce the number of infected cells. “With infections, it’s clear that the system is being misused. If you could cut these connections, you could reduce the number of infections,” Gerdes says. But, he cautions, cutting off links might interfere with cells’ exchange of other critical signals too, which could lead to unexpected problems.
Bodily relevance
So far, nanotubes have been spotted in dishes of human immune cells, brain cells, cancer cells and cells that become heart cells. But seeing something in a dish of cells is not the same as finding it in a whole organism. “The problem is extrapolating from cells hanging out in a dish to the body,” Davis says.
Figuring out what happens (not just what could happen) in an organism is hard to do with dishes of cells. Mothes says, “Most of our science is in vitro. You work with cells that don’t grow into tissue anymore.”
But the cells retain their sticky adhesion molecules on the outside. “It’s a consequence of the inherent desire of cells to form a tissue,” he says. Nanotubes observed between lab-grown cells could be remnants of a collision between two sticky cells with little biological importance, Mothes says.
But Gerdes disputes this idea. “Cells act differently when you put them in a dish, but they don’t invent new things,” he says. To put it another way, hermits may act a little funny but they don’t sprout wings and fly away.
In fact, scientists have found nanotubes connecting immune cells in dissected mouse corneas, suggesting the nanotubes existed in the living mouse eye. These nanotubes increased in number when mice were subjected to stress. The research, led by Paul McMenamin at the University of Western Australia in Perth, is the first example of nanotubes forming in a living, breathing mammal.
For his part, Davis is confident that nanotubes exist in the body. “Being able to prove that they are important in the body is harder to answer,” he says.
Because these hidden communication lines between cells have gone undetected for so long, scientists are struggling with what Gerdes calls the “very basic science” of nanotube form and function. They are just now figuring out the details.
“It’s often that way in science,” Davis says. “You work in the framework of what the field is doing at that time. But it’s the crazy stuff that is going on in the corner that’s exciting.”






