In yet another stab at mimicking nature’s chemical innovations, scientists have devised a new way to make artificial receptors that differentiate among similar molecules.
Chemists have long admired how precisely antibodies select specific chemicals in complex biological brews. This trait has rendered antibodies useful for a broad range of laboratory experiments and medical tests.
Yet antibodies are costly and have a short shelf life, says Steven Zimmerman of the University of Illinois at Urbana-Champaign. Artificial molecules that act like antibodies might avoid these problems, so they may open routes to new medical tests, pollution monitors, and chemical weapons detectors, he says.
Chemists have previously imprinted polymers with shapes that enable them to detect specific molecules. However, the resulting materials each contain many receptor sites, some of which aren’t very selective, says Zimmerman.
In contrast, the new imprinting process developed by Zimmerman and his colleagues at the University of Illinois produces just one receptor site per molecule. The researchers can then discard any molecules with poorly formed sites.
“We have the potential to separate the good from the bad,” Zimmerman says.
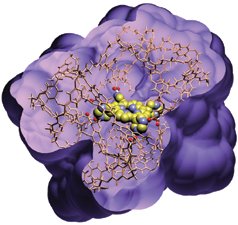
He and his colleagues made their novel receptors by building a large, branched molecule, called a dendrimer, around a template molecule that has a shape similar to that of the molecule they want their artificial receptor to recognize and bind. The resulting dendrimer has a cavity at its center that can bind target molecules.
In the July 25 Nature, the researchers illustrate their technique by using a porphyrin molecule as a template. Porphyrins are large molecules at the heart of such key biological substances as chlorophyll and hemoglobin. To each of these, the researchers bonded eight branched molecules that they then linked into a stable dendrimer molecule. After releasing the central template by breaking the bonds holding it in place, the resulting cavity possessed an imprint resembling the porphyrin.
The researchers found that the cavity would bind five very similar porphyrin molecules but wouldn’t attach to others that carried different numbers of chemical groups.
The new method “is an important step,” comments Andrew Hamilton of Yale University. “It still has to be further elaborated, but I think it’s a clever example of a new approach.”
Zimmerman and his coworkers now are striving to make more-rigid dendrimers that will bind to just one specific molecule. The researchers have also incorporated dye molecules into their dendrimers so that a visible color change occurs when a porphyrin molecule binds, says Zimmerman.
“It’s a very elegant piece of work,” says Jeremy Sanders of the University of Cambridge in England. “It solves many of the problems associated with traditional imprinted polymers, and I think, therefore, it has a lot of potential in various kinds of molecular recognition.”






