New ‘smart’ fibers curb fires in lithium-ion batteries
Tiny capsules release flame retardant when temperatures get too hot
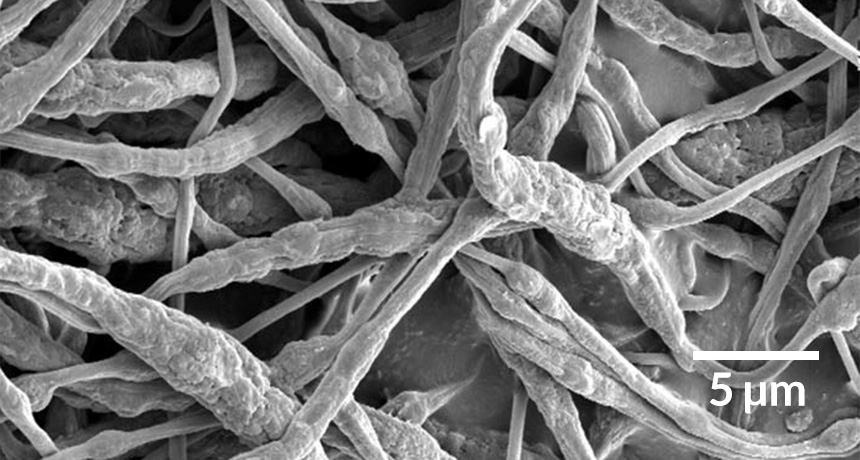
FIRE FIGHTER To stop battery fires in their tracks, researchers formed tiny fibers containing flame retardant, shown above in an image taken by a scanning electron microscope. If the battery overheats, the fibers’ plastic shells melt, releasing the flame retardant encapsulated inside.
Kai Liu et al/Science Advances 2017








