New tests screen for lethal prion disease
Urine and nasal swabs can reveal silent carriers of infectious proteins
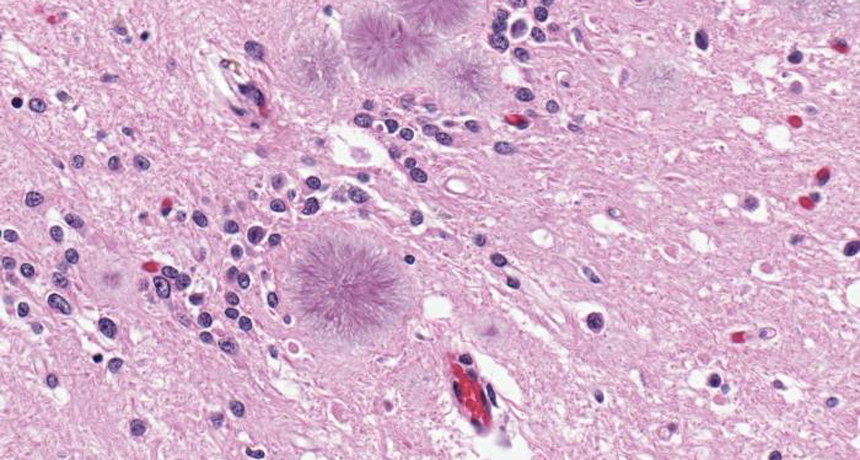
STICK TOGETHER Contagious prion proteins in the brain spawn aggregates (fuzzy purple blobs in this micrograph) that cause Creutzfeldt-Jakob disease. For the first time, noninvasive tests can detect these fatal proteins in urine and nasal tissue.
Sherif Zaki and Wun-Ju Shieh/CDC/Wikimedia Commons