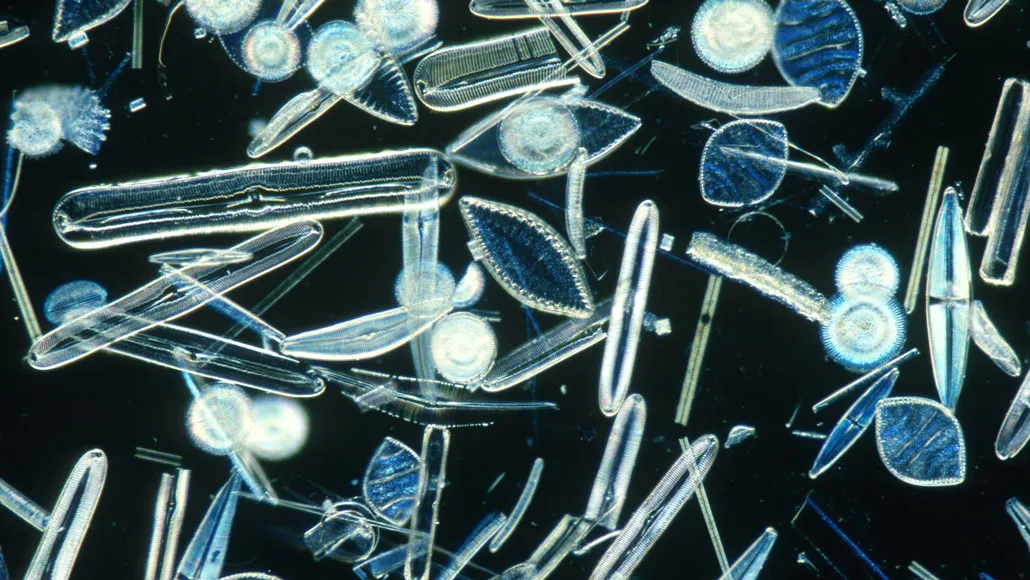
Diatoms’ silica cell walls help drag the tiny carbon-bearing algae down into the ocean when they die. But acidifying oceans can lead to thinner walls.
M.I. Walker/Science Source

Diatoms’ silica cell walls help drag the tiny carbon-bearing algae down into the ocean when they die. But acidifying oceans can lead to thinner walls.
M.I. Walker/Science Source